
Women’s Health: Expanding the Path from Health to Wellness
This series of briefs is directed at Ob/Gyns, Concierge doctors, GPs, Internal Medicine doctors, Integrative Medicine practitioners, Functional Medicine practitioners, Allied Health practitioners, etc. In other words, all those who help women manage their unique physiology. A female’s entire life cycle from menarche through menopause is considered, which also directs these briefs at Pediatricians and Primary Care Physicians.

Table of Contents
Introductory Primer: Parasympathetic and Sympathetic (P&S) Nervous Systems
Women have taken over medicine and women have finally decided to differentiate themselves from men; both as patients and providers and how they're managed. They are “voting with their feet” and changing how medicine (healthcare) is going to be moving forward in the next several years. P&S Monitoring provides more information, enabling this differentiation throughout their entire lifecycle as a curative and a preventative, especially for women treated with HRT but do not feel better, and if anything, are worse.
Understanding the Parasympathetic and Sympathetic (P&S) System
The Autonomic Nervous System (ANS) controls and coordinates every system in the human body. The two branches – the Parasympathetic and the Sympathetic systems – work in cooperation with each other, like the brakes and accelerator of a car; working continuously keep the body in balance. Even if they themselves are out of balance, they work to keep the systems working properly until “the final straw” then symptoms occur. Together, they regulate circulation, metabolism, immune activity, reproductive health, and much more. A woman’s ability to adapt to stress, maintain hormonal rhythm, and sustain fertility depends greatly on how well these two systems remain in equilibrium.

The Parasympathetic System: Rest, Repair, Restore, Protect
The Parasympathetic branch sets the metabolic baseline around which the body reacts. It supports recovery and regeneration. It uniquely controls the Gastrointestinal track motility, Hormones, and the Immune systems; thereby, promoting digestion, nutrient absorption, immune modulation, hormone cycles (growth & development, daily functioning, menstrual cycle), reproductive readiness, and restorative sleep. When functioning optimally, it lowers heart rate and blood pressure, supports menstrual regularity, and stabilizes mood and energy.
Parasympathetic Excess (PE) — or Parasympathetic over-activation — may manifest as chronic fatigue, heavy menstrual flow, lightheadedness and dizziness, and behavioral health dysfunction (including depression/anxiety) due to excessive vasodilation, poor cerebral perfusion and reduced metabolic tone.
The Sympathetic System: Fight or Flight, Stress Response
The Sympathetic branch is the reactionary branch. It mediates the body responses to challenges. It increases heart rate, sharpens alertness, and mobilizes glucose and oxygen for action. In balance, it provides energy and focus; however, Sympathetic Excess (SE) leads to tension, insomnia, Anxiety, elevated blood pressure, and mediates inflammatory and histaminergic responses. In women, prolonged SE is associated with hormonal disruption, hot flashes, and metabolic resistance.
P&S Dysregulation and Women’s Health
Healthy physiology requires a dynamic balance as well – activation followed by recovery. Chronic stress, hormonal shifts, poor sleep, and nutrient deficiencies may disrupt this rhythm, creating P&S dysregulation. Such imbalance contributes to menstrual irregularities, infertility, mood disorders, persistent weight gain, and symptom flare-ups during perimenopause and menopause.
In Women’s Health the P&S network interacts intimately with three key systems:
Endocrine System: modulates estrogen, progesterone, thyroid, and adrenal hormones.
Immune System: influences mast-cell activity, histamine response, and inflammation.
Reproductive System: governs uterine blood flow, ovulation, and pelvic tone.
Visualizing these interconnections highlights how autonomic imbalance serves as a hidden driver behind many “mystery” symptoms in women’s health — linking mind and body, and metabolism, and hormonal rhythms in one integrated framework.
Clinical Implications

Key Points: More information from P&S Monitoring may provide additional protocols that augment hormone replacement therapy (HRT). By also treating autonomic dysregulation, including for example (1) Mast Cell therapy* for abnormal menstrual periods, (2) P&S therapy for post-gestational Behavioral Health issues, and (3) P&S therapy for menopausal symptoms of Night Sweats & Behavioral Health disorders (both of which may be relieved with very low-dose anti-cholinergics, e.g., Nortriptyline, not more than 10.0 mg, qd(7PM)) and Hot Flashes (aka., Vasomotor Syndrome, which may involve dysregulation in both P&S branches.), those who are not responding to HRT may still find relief. Pelvic Floor issues are also associated with P&S dysfunction.
Clinical Takeaway: Mast Cell Activation Syndrome is a type of P&S dysregulation. Hormones are regulated by the Parasympathetics. The vasculature, including Vasomotor Syndrome is regulated by the Sympathetics. Behavioral Health is associated with proper cerebral perfusion which is regulated by P&S function. Sweating and temperature control are regulated by P&S function.
Action Step: All P&S dysfunctions are treatable. Treating P&S dysregulation(s) enhance(s) HRT and improves quality of life and productivity.
Women's Health, Introduction
It's a lifelong challenge, and it may not be all hormonal.
In general, the Parasympathetic nervous system, which is only able to be measured non-invasively and directly by P&S Monitoring, uniquely controls and coordinates the Hormonal, Immune, and Gastrointestinal (GI) systems. It also sets the metabolic threshold around which the Sympathetics react. Abnormal Parasympathetic activity leads to abnormal responses and reactions in all systems of the body, but most directly in the three systems which it uniquely influences. As it turns out, all three of these systems are also key to Women’s Health.
The P&S nervous systems figure prominently in reproductive health, perhaps more so in women than in men. As an aside, in the current culture, it must be stated that significant modifications of reproductive hormones, whether as hormone replacement or augmentation, or as in attempting to change gender, will significantly impact P&S function and thereby physical and mental health and well-being. The Parasympathetic nervous system, in part, controls and coordinates the hormones in the body, especially the Thyroid and Reproductive hormones, directly, and the Reproductive hormones indirectly through the Thyroid as well. In fact, in women, at the level of the reproductive organs, Estrogen and Progesterone are neuromodulators and neurotransmitters helping the nervous system communicate with the reproductive organs. So, more hormones, more ability to communicate, is not always the best treatment, especially if excess communication is already the problem, resulting in abnormal periods for example.
Oxidative Stress has been identified to play a key role in the pathogenesis of subfertility in both males and females. The adverse effects of Oxidative Stress on sperm quality and functions have been well documented. In females, on the other hand, the impact of Oxidative Stress on oocytes and reproductive functions remains unclear. Oxidative Stress may lead to a number of reproductive diseases, including Endometriosis, Polycystic Ovary Syndrome (PCOS), and unexplained infertility. Pregnancy complications, such as spontaneous abortion, recurrent pregnancy loss, and preeclampsia, may also develop in response to Oxidative Stress [1]. Oxidative Stress is a major contributor to P&S dysregulation.
Mast Cell over activation may also lead to a number of reproductive diseases, including Dysmenorrhea, Endometriosis, Fibroids, and Polycystic Ovary Syndrome (PCOS) [2]. Mast Cells are immune cells and, as with Hormones, are also controlled and coordinated by the Parasympathetic nervous system. The release of Mast Cells is by the Parasympathetics and the release of Histamine from Mast Cells (aka, Degranulation) is mediated by the Sympathetics. As with Oxidative Stress, P&S dysregulation is also involved in Mast Cell Activation Syndrome, contributing to Women’s Health issues. The good news is that all P&S dysfunctions are treatable.
This series of briefs will provide more insight into the relationship between the P&S nervous systems and Women’s health, from Menarche to Menopause. We know you are not just small men. We hope to help women take more control over their healthcare and continue to discover new ways to help you achieve wellness and stay well.
Clinical Implications

Key Points: P&S balance, specifically normal Parasympathetic activity, is required for normal hormone and Mast Cell regulation.
Clinical Takeaway: Hormone imbalance and Psychological disorder may not be all that is involved in the “mystery” symptoms in women’s health.
Action Step: Screen for P&S dysfunction for more information and treat the whole person as an individual.
Uro-Gynecology & Bowel Overview, Part 1
The P&S nervous systems control and coordinate, as well as integrate, all systems of the body. Through the P&S systems, Women’s Health issues are linked to many other systems of the body, including behavioral health, gastrointestinal health, immune health, cardiovascular health, and more.
Women’s pelvic region, including the bladder, uterus, bowels, and a host of pelvic supporting ligaments, is a site of frequent and often excessive activity, leading to a host of urogynecological and lower bowel disorders, as well as behavioral health issues especially around the time of a woman’s period. This is caused by excessive vasodilation due to the fact that estrogen and progesterone are vasodilators and are more abundant at that time. Vasodilation, if exacerbated by P&S dysregulation, leads to poor cerebral perfusion, which may lead to sub-clinical or clinical depression, which may lead to anxiety or anxiety-like symptoms, bipolar, headache or migraine, lightheadedness, brain fog, fatigue, sensory, memory, cognitive, or sleep difficulties.
For example, in cases of heavy, irregular, prolonged, or otherwise abnormal flows, more hormones typically are not always the better solution. Like the rest of P&S dysfunction, less is the better treatment, especially since Progesterone and Estrogen are vasodilators, making it harder for the heart to pump blood to the brain and easier to pump more blood to the pelvic region. In fact, the Parasympathetics communicate with the reproductive organs through the reproductive hormones. If the Parasympathetics are already over-communicating, prescribing more reproductive hormones is counter-intuitive.
Urogynecological and lower bowel disorders may include vulvar or pelvic floor symptoms, such as swelling of the labia, prolapsing of an internal organ (the patient might feel a bulge in the vagina from a uterine or bladder prolapse, for example), or Persistent Genital Arousal. Among the mechanisms believed to be involved in these symptoms are excessively dilated veins in the pelvic region, which results in pooling of blood, and lax ligaments (perhaps due to large or multiple pregnancies), which normally bolster organs like the bladder and uterus, but whose “floppiness” instead causes their collapse. Both of these mechanisms may also cause compression of nerve branches and involve pain beyond the menstrual cycle and perhaps exacerbated during the menstrual cycle due to increases in Progesterone which tends to increase the laxity of connective tissue.
Hormones, Oxidative Stress, and P&S dysfunction contribute to Lower Urinary Tract Symptoms including: urinary urgency, increased urinary frequency with or without incomplete emptying of the bladder, straining to empty the bladder, urinary retention, a weak urinary stream, urge incontinence, night-time incontinence, and increased night-time urination (nocturia). These symptoms may be exacerbated by MCAS. Treating more than one possible cause often leads to better outcomes.
Overall, the pelvic region of a female body is a very active and potentially very stressed area of the female body, and if not properly regulated and managed by the various systems of the body that effect it, and properly controlled and coordinated by the P&S nervous systems, a woman’s health, including quality of life and productivity, is severely affected.
Clinical Implications

Key Points: P&S balance underpins hormonal, immune, and GI regulation.
Clinical Takeaway: Routine P&S monitoring may help detect early dysautonomia that precedes hormonal or metabolic imbalance in women presenting with fatigue, menstrual irregularities, or mood fluctuations.
Action Step: Screen for P&S dysfunction before initiating hormone therapy or antidepressants.
Uro-Gynecology & Bowel Overview, Part 2
Among all systems, the Parasympathetics also directly control and coordinate the Immune system. Mast Cells are immune cells. All surfaces of the body that interface with the outside world are replete with Mast Cells. Increased Parasympathetic activity during menstruation increases Mast Cells reactivity, which may lead to greater blood flow, greater inflammation, and greater pain. Fortunately, there are approaches that individuals may take to regulate and stabilize symptoms. Understanding these issues early in life may lead to effective interventions that may dramatically improve the course of a young woman’s reproductive years and beyond.
Oxidative Stress is a persistent and ubiquitous issue with uterine health, which is compounded by the other environmental, social, mental, emotional, psychological, physical, and physiological stresses. This information may be scary. We understand. The good news for this is that Oxidative Stress is treatable.
Oxidative Stress (e.g., due to Reactive Oxygen Species or Reactive Nitrogen Species) has been identified to play a key role in the pathogenesis of subfertility in females; however, the impact of Oxidative Stress on oocytes and reproductive functions remains unclear. Oxidative Stress may lead to a number of reproductive diseases, such as Endometriosis, Polycystic Ovary Syndrome (PCOS), and unexplained infertility. Pregnancy complications such as spontaneous abortion, recurrent pregnancy loss, preeclampsia, and pre-term labor may also develop in response to Oxidative Stress. Studies have shown that extremes of body weight and lifestyle factors such as cigarette smoking, alcohol use, and recreational drug use promotes Oxidative Stress, which could affect fertility, as well as pregnancy [1]. Excessive exposures to environmental pollutants (e.g., pesticides, lead, Polychlorinated Biphenyls, and other air, soil, and water pollutants) and excessive consumption of processed foods, both from the processing additives and the processing itself (removing nutrients) [1], are of increasing concern, as they too have been found to trigger oxidative states, possibly contributing to female infertility and pregnancy. [1]
The main P&S dysregulation that underlies this, Parasympathetic Excess (PE), was initially discovered in women who have had more than two pregnancies. In these cases, pregnancy is defined as a pregnancy that lasts at least through the first trimester before termination. The symptoms attributed to motherhood are included in the symptoms of PE. As a result, it was originally going to be called “Motherhood Syndrome.” However, we started to find it in men and couldn't call it “Motherhood Syndrome” anymore.
PE may be due to many causes. PE will affect all stages of a woman’s life. For example, women with PE typically start menopause early (perimenopause) or have miserable menopause with more frequent and greater night sweats, hot flashes, and behavioral health issues. Relieving PE has been found to help relieve night sweats and behavioral health issues and lessen hot flashes (suggesting that hot flashes are not all Parasympathetically mediated, as suggested by the clinical title of hot flashes – Vasomotor Syndrome. The “motor” in the title implicates the Sympathetic nervous system).
As seen above, as we go deeper into the control and coordination of a woman’s pelvic region, more information becomes available to help manage an individual’s women’s health, to maintain quality of life and productivity, potentially cure some of these issues, and help women to wellness. Women’s physiology is distinct – women are not simply smaller versions of men. Each individual’s biology is shaped by unique patterns of P&S activity. Restoring balance within these systems is foundational to reestablishing women’s overall health and resilience. The good news is that the source of the more information, P&S activity, is also what helps to make everyone different. Restoring P&S health is foundational to restoring Women’s Health.
Clinical Implications

Key Points: Excess vasodilation from estrogen/progesterone, amplified by P&S imbalance, affects pelvic blood flow and cognition.
Clinical Takeaway: Treating P&S dysregulation may reduce pelvic congestion, urinary urgency, and cognitive symptoms often mislabeled as “psychogenic.”
Action Step: Incorporate autonomic assessment alongside pelvic floor evaluation.
Menarche, Menstrual Issues, Part 1: Oxidative Stress
Oxidative Stress is a persistent and ubiquitous issue with uterine health, which is compounded by the other environmental, social, mental, emotional, psychological, physical, and physiological stresses. Oxidative stress is also a common and ongoing concern in uterine health, often intensified by environmental, social, emotional, psychological, and physical factors. While this may sound concerning, there’s encouraging news – oxidative stress may be identified, managed, and effectively reduced with the right interventions.
Oxidative Stress (e.g., due to Reactive Oxygen Species, ROS, or Reactive Nitrogen Species, RNS) has been identified to play a key role in Women’s Health. Oxidative Stress may lead to a number of reproductive diseases such as Dysmenorrhea, Endometriosis, and Polycystic Ovary Syndrome (PCOS). Studies have shown that extremes of body weight and lifestyle factors such as cigarette smoking, alcohol use, recreational drug use, and excessive workouts (as in the case of athletes with dysmenorrhea) promotes Oxidative Stress, which could affect menstruation [1]. Excessive exposures to environmental pollutants (e.g., pesticides, lead, Polychlorinated Biphenyls or PCBs, and other air, soil, and water pollutants) and excessive consumption of processed foods, both from the processing additives and the processing itself (removing nutrients) [1], are of increasing concern, as they too have been found to trigger oxidative states.
Antioxidant supplementation may be effective in controlling Oxidative Stress [3]. Exercise is arguably the most potent of antioxidants, including exercise during pregnancy [4, 5, 6, 7]. The most potent anti-oxidant that heals the nervous system is r-Alpha-Lipoic Acid (600 mg, tid, or equivalent). Note, this is what the body already makes, and it may be supplemented as the natural production falls off due to aging or chronic disease. However, a certain amount of ROS is needed for Immune health and the progression of normal cell functions, provided that upon oxidation, every molecule returns to its reduced state [8]. Excessive Reactive Oxygen Species production, an antioxidant-oxidant imbalance, for example, may overpower the body’s natural antioxidant defense system, creating an environment unsuitable for normal female physiological reactions (see Figure 1).
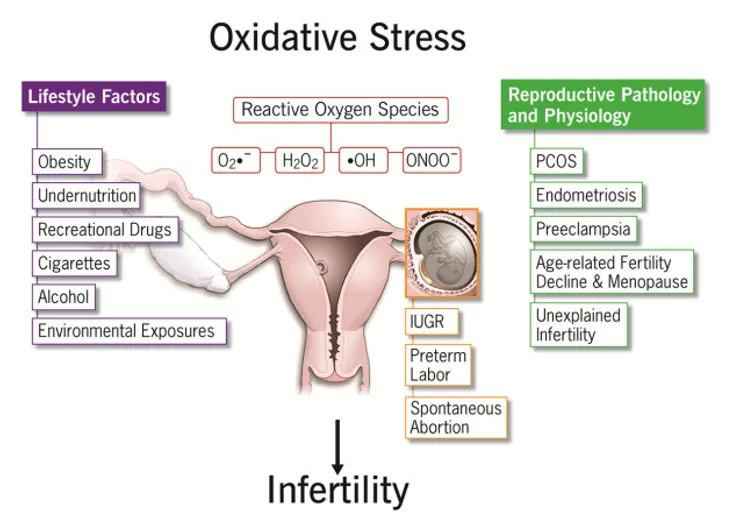
Figure 1
Factors contributing to the development of oxidative stress and their impacts on female reproduction. See text for more details.
Clinical Implications

Key Points: Oxidative Stress plays a key role in Women’s Health.
Clinical Takeaway: Antioxidants not only helps to restore P&S balance, it also helps to relieve oxidative stress. The two most potent anti-oxidants the body makes are r-Alpha-Lipoic Acid (600 mg, tid, or equivalent) which heals the nervous system, and CoQ10 (200 mg, qd) which heals the heart and vasculature. Note, these are what the body already makes, and the may be supplemented as the natural production falls off due to aging or chronic disease.
Action Step: Establish and maintain a proper oxidant/antioxidant balance.

Menstrual Issues, Part 2: Mast Cells, Dysmenorrhea
In Women’s Health, the Uterus is one of the barriers between the woman and the outside world. It is normally and naturally replete with Mast Cells. The Mast Cells in the Uterus are released during the monthly cycle. With Parasympathetic Excess (PE), too many Mast Cells are released. PE also causes secondary Sympathetic Excess (SE), which releases too much Histamine from each Mast Cell, resulting in an order of magnitude increase in Histamine release. Like in a bug bite, Histamine draws inflammation. As a result, inflammation is amplified. Inflammation is further amplified by the secondary SE, as a direct effect. The excessive inflammation draws excessive amounts of blood. This process is often the cause of prolonged or excessive flows and blood clots. Collectively, these excesses may also be the cause of Dysmenorrhea, and, in conjunction with the connective tissue dysfunction, Endometriosis, Polycystic Ovary Syndrome, connective tissue over-growth causing cysts and fibroids, and other related disorders. Many of these hormone-related disorders are exacerbated by the hormone related inflammation that coincides. Again, relieving PE (involved in both the hormone-exaggerated response and the amplified inflammatory response) while relieving base inflammation helps to relieve these disorders. Mast Cells also carry receptors for reproductive hormones and may be the cause of unexplained allergies, chemical sensitivities, or asthma, as well as additional inflammation [9]. Fibroids, depending on location, may also cause uterine bleeds as well as more pain, and the additional bleeding may lead to iron deficiency, thereby compounding problems [10]. Don’t forget Progesterone. It normally increases during the luteal phase, or the second half of the menstrual cycle. [11]. Progesterone tends to make connective tissue in general more stretchy, which may lead to increased permeability of the Uterus promoting Endometriosis and thereby possibly PCOS.
Dysmenorrhea may have many causes. It and associated menstrual disorders may be exacerbated or made more frequent by the laxity or dysfunction of connective tissue due to, for example, large or multiple pregnancies. Generally, it is a problem with muscle contractions of the Uterus. The connective tissue that surrounds and facilitates uterine muscle contractions may be dysfunctional due to adhesions, overgrowth, prolapses, scar tissue build-up, Hormone or P&S imbalance, etc. This may be compounded by heavy and prolonged flows. Often, Dysmenorrhea and Endometriosis are interchanged or misdiagnosed as the other. Fortunately, the treatment tends to be the same.
Again, Mast Cells are immune cells and the Parasympathetics control and coordinate the Immune system. While PE is being relieved, MCAS may be managed to improve quality of life and productivity. To understand treatment, note that PE releases too many Mast Cells and from the Mast Cells, Histamine is released. Histamine release may be blocked, but both main Histamine pathways (H1 & H2) must be blocked to be effective. If only one path is blocked, the body will use the unblocked pathway. This is why both must be blocked, for example, from Zyrtec 10 mg/day for H1 and Pepcid 20 mg/day for H2. If Histamine blockers are not effective or tolerated, then the Mast Cells themselves may be sequestered. However, often it takes prescription-grade medications
Clinical Implications

Key Points: The Uterus is normally and naturally replete with Mast Cells.
Clinical Takeaway: PE releases too many Mast Cells and causes secondary SE which releases excessive amounts of Histamine which draws too much blood. Histamine draws inflammation which is amplified by SE. Excessive inflammation draws more excessive amounts of blood. This process is often the cause of Dysmenorrhea, and may lead to Endometriosis, Polycystic Ovary Syndrome, cysts and fibroids, and other related disorders.
Action Step: Relieve PE, and in the meantime, normalize Mast Cell activity.
Menstrual Issues, Part 3: Endometriosis
Endometriosis is known as a benign, estrogen-dependent disorder characterized by the presence of endometrial tissue outside the uterus and is known to be associated with pelvic pain and infertility [12]. As we mentioned, MCAS releasing too much Histamine and thereby drawing more inflammation may also promote Endometriosis. The peritoneal fluid of patients has been found to contain high concentrations of pro-inflammatory cytokines (IL-6, TNF-alpha, and IL-beta), angiogenic factors (IL-8 and VEGF), monocyte chemoattractant protein-1, and oxidized low-density lipoproteins [13, 14]. Pro-inflammatory and chemotactic cytokines play a central role in the recruitment and activation of phagocytic cells, which are the main producers of Oxidants, both ROS and RNS [15]; to name a few.
Heat Shock Proteins function to help maintain normal processes, such as protein folding and translocation, and promote cell proliferation, while maintaining quality control [16, 17, 18, 19, 20]. They are present in low levels under normal conditions [21] and in high levels under situations of stress [1357]. Under the latter conditions, Heat Shock Proteins promote Oxidative Stress, stimulating more inflammation, possibly accounting for pelvic inflammation and growth of endometriotic tissue [1363, 22, 23, 24]. Oxidants are thought to encourage growth of ectopic endometrial tissue through the induction of cytokines and growth factors [25] (see Figure 2). Oxidative Stress stimulates inflammation, invasion, angiogenesis, and cell proliferation, and prevents limitations on the growth (apoptosis, aka programmed cell death) of endometriotic cells [26]. N-acetylcysteine (NAC) and Vitamin E are antioxidants that limit the proliferation of endometriotic cells [27, 28].

Iron mediates production of Oxidants (Reactive Oxygen Species), thereby inducing Oxidative Stress [29] (Figure 2). In the peritoneum of patients with endometriosis, accumulation of iron and cells containing iron (heme) around endometriotic lesions [30] from retrograde menstruation (blood flowing back up the Fallopian tubes) [31] and increases the generation of Nitric Oxide [32], but also more Oxidants. Naturally, endometriotic cysts contain high levels of free iron. Chronic oxidative insults from iron buildup within endometriotic lesions may be a key factor in the development of the disease [33]. There are several proposed mechanisms leading to endometriosis in addition to MCAS, Oxidants/Antioxidants imbalance, and Inflammatory processes, including those that involve iron and retrograde menstruation.
Patients with Endometriosis tend to have lower pregnancy rates than women without the disease. Low oocyte and embryo quality, in addition to environments toxic to sperm preventing conception, all contributing to the subfertility experienced by patients with Endometriosis [34]. The peritoneal fluid of women with Endometriosis contains low concentrations of antioxidants proposed to be secondary to decreased Progesterone response of Endometrial cells [35]. Diets lacking adequate amounts of antioxidants may predispose some women to Endometriosis [36, 37, 38, 39, 40, 41, 42, 43, 44].

Figure 2: Malignant transformation of Endometriosis: a fine-tuned balance between oxidants and antioxidants. Cell-free hemoglobin, heme, and iron massively released into the endometriotic cyst fluid space during menstruation are prone to self-oxidation (autoxidation) and may spontaneously convert (normal) oxygenated Hemoglobin (oxyHb) to Hemoglobin that may no longer bind oxygen (metHb). Reactive Oxygen Species (ROS, O2–) are continuously generated by the autoxidation of Hemoglobin. Iron derivatives also stimulate the generation of additional ROS (OH) in endometriotic cysts. Furthermore, Hemoglobin and Heme stimulate antioxidants. Antioxidants are considered to be a double-edged sword. Excess ROS cause cell death. Antioxidants alleviate cell death by neutralizing ROS, allowing for increased cell survival. [45, Open Access]
Endometriosis impacts reproduction, health, and quality of life. It is highly Estrogen-dependent. It is well associated with high levels of chronic stress. The stress intensity correlates with pain severity and disease extension. However, it is unknown whether chronic stress represents a primary cause of Endometriosis or Endometriosis adds to the stress levels [46]. It is likely both.
Clinical Implications

Key Points: Oxidative stress and Mast Cell activation with PE exacerbate dysmenorrhea and mood disorders and may promote Endometriosis.
Clinical Takeaway: Targeting oxidative load (diet, antioxidants, e.g., r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent), stabilizing Mast Cell activity (quercetin, vitamin C), and anti-inflammatories may improve cycle regularity.
Action Step: Use integrative therapies that support antioxidant balance and autonomic tone.
Menstrual Issues, Part 4: Treatment for Endometriosis
While there is no known cure for Endometriosis at this time, stress reduction, antioxidants, and anti-inflammatories are highly suggested to help relieve some of the painful symptoms of Endometriosis, including dysmenorrhea (painful periods), pain during intercourse (dyspareunia), or pain during bowel movements or urination. Lately we also find that, if MCAS is involved, antihistamines or Mast Cell sequesters may also help. Of course, (1) stress, including Oxidative Stress, (2) inflammation, including through excessive histaminergic responses, and (3) P&S dysfunction that amplifies the first two as well as the pain are also involved. Further, the laxity of the connective tissue in the pelvic region, including from multiple or heavy pregnancies or connective tissue disorders (e.g., Hypermobility) may promote menstrual blood, containing endometrial cells, to “leak-out” of the uterus. In general, hormone therapy and reducing Oxidative Stress, currently, seem to be the primary therapy options for Endometriosis. However, this may not be all.
Hormone Therapy (unless pregnancy is soon desired) to reduce the number and size of growths (implants) and keep them from spreading
Estradiol to reduce the higher levels of endogenous Estrogen produced by the Ovaries in women with Endometriosis and to regulate how uterine tissue grows to prevent it from growing outside the uterus
Progestin to reduce menstrual flow especially for women who may not take Estrogen because of cardiovascular disease or a high risk of blood clots
Stress Reduction to reduce Oxidative stress through
Antioxidants, including mild exercise (exercise is arguably the most potent of antioxidants), Zinc, r-Alpha Lipoic Acid, or Co-Q10
Anti-inflammatories, including (1) Turmeric – Curcumin, the active ingredient in Turmeric, which may inhibit the growth of endometrial cells, (2) Omega-3 Fatty Acids with fewer Omega-6 Fatty Acids, and (3) Vitamin D, which may reduce inflammation, reduce the spread of cells, and reduce the formation of blood vessels to lesions, which as a result, may control the occurrence and growth of Endometriosis
Vitamin D, which is also an anti-inflammatory, is additionally recommended since apparently women with Endometriosis have lower vitamin D levels than normal [47] and Vitamin D deficiency may be a potential risk factor for Endometriosis
Systemic Stress Reduction (which will also reduce Oxidative Stress), including improved restorative sleep, reduced alcohol and inflammatory food intake, avoiding known (stress) triggers for the individual, hot compresses, and mild exercise
Histamine Blockers to reduce the effects of Mast Cells: H1 blocker e.g., Zyrtec 10 mg daily, and H2 blocker e.g. Pepcid 20 mg daily.
Mast Cell Sequesters (e.g., Cromolyn) to reduce MCAS by preventing Mast Cell release to begin with or reduce the inflammatory effects of Mast Cells.
Ultimately, Laparoscopic surgery to remove the endometriotic tissue helps reduce pain, but pain often returns within a year or two; however, removing the ovaries or uterus usually relieves pain
Clinical Implications

Key Points: There may be more to Endometriosis than currently accepted
Clinical Takeaway: Hormone therapy and reducing Oxidative Stress seem to be the primary therapy options. However, this may not be all.
Action Step: Monitoring P&S activity, inflammatory and histaminergic markers may provided more information to aid therapy planning.
Menstrual Issues, Part 5: Polycystic Ovary Syndrome (PCOS)
Polycystic Ovary Syndrome (PCOS) is also a very common hormone problem for women of childbearing age. It has a prevalence of about 18% [48]. Women with PCOS have ovulatory dysfunction and polycystic ovaries (see insert, right [49]). They may not ovulate or have missed, few, unusual, irregular, or very long menstrual periods (variable periods during which the flows range from absent periods to heavy or prolonged periods). They may have many small cysts on the ovaries. They may also have high levels of androgens (a male hormone, known as hyperandrogenism), with excess hair growth (Hirsutism), skin disorders (e.g., Acne), infertility (up to 90% of women with PCOS cannot conceive), and weight gain. PCOS causes many small sacs of fluid to develop on the ovaries, preventing them from regularly releasing eggs.
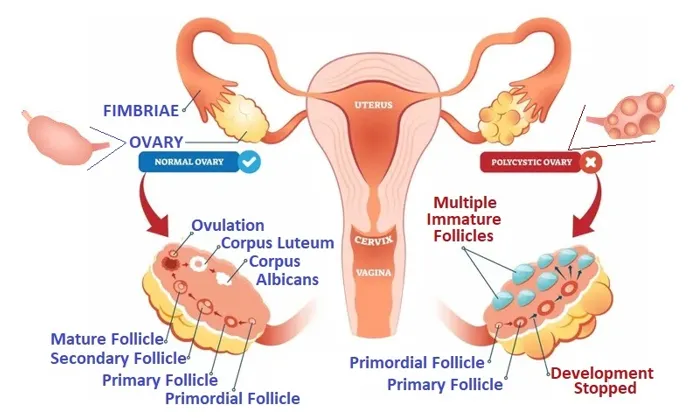
As discussed, MCAS in Women’s Health may also lead to heavy, irregular flows with blood clots, similar to PCOS. It may be that histamine blockers may help to control some symptoms of PCOS through better management of MCAS.
Insulin resistance may be involved in PCOS. Signs of insulin resistance such as hypertension, obesity, and central fat distribution are associated with insulin resistance [50]. Insulin resistance, or compensatory Hyperinsulinemia, increases the availability of both circulating Androgen and Androgen production which are, in part, characteristic of PCOS [51].
PCOS is associated with decreased antioxidant concentrations and increased Reactive Oxygen Species production causing mitochondrial dysfunction; as a result, PCOS is considered an oxidative and thereby an inflammatory state [52, 53, 54]. The resultant Oxidative Stress creates an inflammatory environment that further increases Insulin resistance and contributes to Hyperandrogenism [55].
The Parasympathetic system communicates with a woman’s reproductive organs through hormonal signaling, influencing functions such as ovulation, uterine tone, and overall reproductive balance. Parasympathetic Excess (PE) is a condition where the Parasympathetics are already over-communicating. More reproductive hormones will only increase this over-communication. Supplement and lifestyle modification treatment for women with PCOS is known to be helpful. This includes antioxidants (r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent), anti-inflammatories (Turmeric (Curcumin), up to 1000 mg, tid), exercise, and a balanced diet, with a focus on caloric restriction to counter any insulin resistance [56]. However, if lifestyle modifications do not suffice, a variety of options for medical therapy exist. Currently, there is no clear primary treatment for hirsutism, although it is known that combination therapies seem to produce better results [55]. Combined oral contraceptives are considered the primary treatment for menstrual disorders. However, as said before, with P&S dysfunction, more is not a better treatment. Even with hormone therapy, the lower the dosage, the better, titrated to normalizing the underlying P&S dysfunction contributing to PCOS.
Clinical Implications

Key Points: PCOS is considered an oxidative, and thereby, an inflammatory state
Clinical Takeaway: Targeting decreased anti-oxidant concentrations and mitochondrial dysfunction may relieve or prevent PCOS
Action Step: Establish and maintain P&S and Antioxidant balance (r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent), and reduce inflammation (exercise and Turmeric (Curcumin), up to 1000 mg, tid).
Hormones and Women’s Health
More hormones is not always better. It is well known that the Parasympathetic nervous system controls and coordinates all hormones throughout all female lifecycles. In general: Pituitary (growth) hormones and the Parasympathetics work together to control and coordinate development, including the Menarche. Pituitary hormones are the “master control” hormones for the brain and the body, and work with the P&S systems within the brain and brain stem. Thyroid hormones are the “master control” hormones outside the brain and, together with the P&S nervous systems, have direct influence over daily and monthly reproductive hormone activity.
Thyroid Hormones
It is well known that P&S dysfunction in abnormal Thyroid function is common. Most of the cardiovascular and neurological manifestations of abnormal Thyroid function are due to P&S dysfunction. For example, in hypothyroidism, the published major findings are that Sympathetic dysfunction was observed more as compared to Parasympathetic dysfunction [57]. While we see this with most standard forms of autonomic monitoring, it is well known that the Parasympathetics control the hormones. With Cardio-Respiratory monitoring, we find that Parasympathetic Excess (PE) in response to Sympathetic challenges causing secondary Sympathetic Excess (SE) is highly correlated with hormone abnormalities, including Hypothyroidism. Furthermore, our clinical evidence has shown that a significant reduction in Thyroid-hormone replacement therapy is associated in most patients with relief of PE. Therefore, PE promotes Thyroid dysfunction, and in turn, Thyroid dysfunction promotes dysfunction in other hormone functions, especially reproductive hormones, especially in females.
Reproductive Hormones
Hormones and Histamine
The uterus is loaded with H1 (Histamine receptors)
Estrogens trigger mast-cell degranulation
Testosterone and other androgen are credited to having a stabilizing effect
Progesterone has also been shown to have a stabilizing effect on mast cells.
Hormone balance can have profound impact on histamine intolerance/MCAS symptoms
Mast Cells are present in all tissues that interface with the outside world. The uterus is another barrier between a woman and the outside world. Mast Cells are part of the immune and neuroimmune systems. The Parasympathetics release Mast Cells and the Sympathetics release the Histamine from each Mast Cell (known as Degranulation). The uterus is replete with H1 Histamine receptors and Mast Cells. Histamine symptoms are more common in women and often track with the menstrual cycle. They occur when Estrogen is high at ovulation and then again just before the period or when women take the contraceptive pill or hormone replacement therapy (women may feel worse when starting therapy due to Histamine intolerance).
The connection between Mast Cells, Histamine, and hormones is that Estrogen stimulates Mast Cell degranulation (Histamine release from Mast Cells) and down-regulates the Diamine Oxidase1 enzyme that clears Histamine. At the same time, Histamine stimulates the Ovaries to make more Estrogen. PE amplifies each step in this process and causes (beta) Sympathetic Excess (PE) which amplifies Histamine release, leading to an order of magnitude increase in Histamine. The net result may be a vicious cycle exacerbating the blood flow. The excess Histamine leads to more inflammation which in turn draws more blood, and the inflammation is also amplified by the SE caused by the PE. Then Progesterone is added to the cycle. Progesterone is a vasodilator helping to bring even more blood to the area. As a result, heavy and prolonged flows result, leading to cramping and Dysmenorrhea, etc. The presence of this more blood increases the chances of the blood diffusing back into the abdomen, causing Endometriosis. The body does not like blood outside blood vessels and wraps this excess blood in connective tissues, causing fibroids, which, if they occur near the ovaries, cause PCOS. Excessive Histamine release, coupled with the PE, may also promote digestive issues, fatigue, acid reflux, weight gain, hair loss, trouble sleeping, and anxiety. If this is the case, while PE (the root cause of this non-hormonal reason for heavy, prolonged, or abnormal, monthly flows) is being relieved to stop the entire cascade, Histamine blockers (blocking both H1 and H2 pathways) or Mast Cell sequestering medicines may help improve quality of life and reduce the risk of Endometriosis and PCOS, in the meantime. Relieving PE, relieves the entire cascade, as the long-term remedy.
1 Diamine Oxidase is a digestive enzyme produced in your kidneys, thymus, and the intestinal lining of your digestive tract. Its primary function is to break down excess histamine in your body.
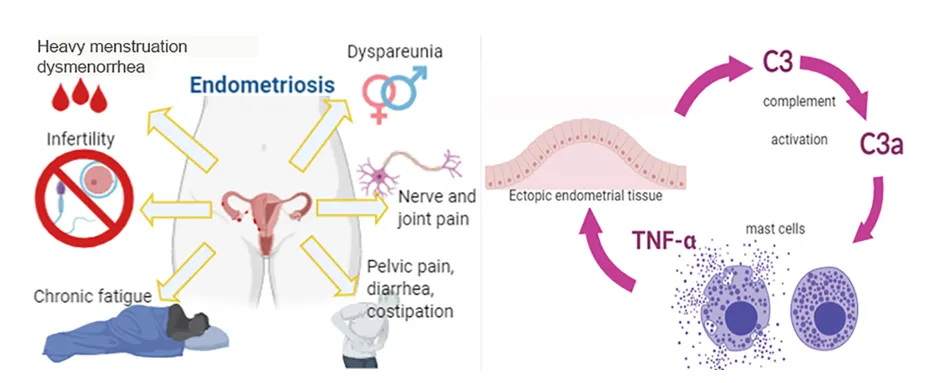
Figure 2: C3a from C3 is a powerful pro-inflammatory mediator that promotes inflammation by attracting immune cells and stimulating cells like Mast Cells. C3 is from the “Compliment System” which is part of the immune system. The complement system, when activated, triggers cascades to identify and destroy pathogens, clear debris, and promote inflammation. There is evidence that C3a participates in an auto-amplifying loop leading to Mast Cell infiltration and activation.
‘Estrogen dominance’ happens when you have more Estrogen than Progesterone. Many symptoms of ‘Estrogen dominance’ are actually symptoms of Histamine or Mast Cell activation. For example, Mast Cells and Histamine play a role in both Endometriosis and Premenstrual Dysphoric Disorder. It is important to note that Estrogen dominance doesn’t just happen with high Estrogen. Even with low Estrogen, Estrogen may be dominant as long as there is more Estrogen than Progesterone. Hormone balance (i.e., Progesterone and Testosterone) has a profound effect on Histamine intolerance and Mast Cell Activation Syndrome (MCAS) symptoms. For example, Progesterone helps stop Mast Cells from making Histamine. Hormone balance and P&S balance stabilizing Mast Cells response leads to normalizing Histamine responses. Reproductive hormones, which reduce nearing menopause, may cause more intolerances and allergies.
Again, women tend to have more Histamine than men because women have more Estrogen. Estrogen, Progesterone, and Histamine are closely linked in the body. Estrogen stimulates Mast Cells to make more Histamine. This may be why women may experience more Histamine issues at certain times in their cycle – likely when Estrogen levels are higher than Progesterone levels. PE and SE tend toward the more symptomatic end of the spectrum due to Mast Cell over-production due also to PE exacerbated by overly-excessive Histamine release (Degranulation) due also to SE caused by the PE.
Again, women tend to have more Histamine than men because women have more Estrogen. Estrogen, Progesterone, and Histamine are closely linked in the body. Estrogen stimulates Mast Cells to make more Histamine. This may be why women may experience more Histamine issues at certain times in their cycle – likely when Estrogen levels are higher than Progesterone levels. PE and SE tend toward the more symptomatic end of the spectrum due to Mast Cell over-production due also to PE exacerbated by overly-excessive Histamine release (Degranulation) due also to SE caused by the PE.
Clinical Implications

Key Points: In addition to the P&S and Immune systems, and reproductive hormones, let us not forget Thyroid hormones.
Clinical Takeaway: Thyroid dysfunction, including due to PE, may also contribute to menstrual dysregulation through all of the above means.
Action Step: Establish and maintain proper Thyroid function.
Conception (Fertility) Issues – Oxidative Stress
Oxidative Stress has been identified to play a key role in fertility in females, including unexplained infertility. However, the impact of Oxidative Stress on oocytes and reproductive functions remains unclear. Pregnancy complications such as spontaneous abortion, recurrent pregnancy loss, preeclampsia, and pre-term labor may also develop in response to Oxidative Stress. Studies have shown that extremes of body weight and lifestyle factors such as cigarette smoking, alcohol use, and recreational drug use promotes Oxidative Stress, which could affect fertility, as well as pregnancy [1]. Excessive exposures to environmental pollutants (e.g., pesticides, lead, Polychlorinated Biphenyls, PCBs, and other air, soil, and water pollutants) and excessive consumption of processed foods, both from the processing additives and the processing itself (removing nutrients) [1], are of increasing concern, as they too have been found to trigger oxidative states, possibly contributing to female infertility and pregnancy. [1]
Antioxidant supplementation (e.g., r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent) may be effective in controlling Oxidative Stress and continues to be explored as a potential strategy to overcome reproductive disorders associated with infertility [58]. Exercise is arguably the most potent of antioxidants, including exercise during pregnancy [59, 60, 61, 62]. Of course, exercise needs to be balanced by proper diet, including food with healing benefits (nutraceuticals).
Oxidative Stress impacts Assisted Reproductive Techniques, and antioxidant supplementation may increase the likelihood for success [63, 64]. However, a certain amount of Reactive Oxygen Species (ROS) is needed for Immune health and the progression of normal cell functions, provided that upon oxidation, every molecule returns to its reduced state [65]. Excessive ROS production, for example, may overpower the body’s natural antioxidant defense system, creating an environment unsuitable for normal female physiological reactions (see Figure 1 in Menarche).
Oxidative Stress also directly affects the nervous system by depleting Mitochondrial activity, thereby reducing the energy required by nerves to work. This includes the P&S nervous systems. Reduced P&S function not only affects brain and heart function but also affects hormone activity. The Parasympathetics communicate with the reproductive organ through the reproductive hormone. If the Parasympathetics are under-active, this also dysregulates hormone activity and will affect the menstrual cycle, ovulation, and possibly the proper environment for conception.
Clinical Implications

Key Points: Unexplained infertility may mean more information is needed.
Clinical Takeaway: Oxidative Stress impacts Assisted Reproductive Techniques. This includes Oxidative Stress due to pathogens (viruses, bacteria, molds, and mildews) and from whole body stressors (emotional, physical, physiological, psychological, mental, social, hormonal, and even excessive healthy stresses like too much exercise).
Action Step: Antioxidant supplementation (e.g., r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent) may increase the likelihood for success.
Conception (Fertility) Issues – Comfort vs. Pain
Conception may be made more difficult if the looseness of the connective tissue in the vaginal walls creates more friction and discomfort. This is due to the tissue being loose and made to wrinkle during intercourse, as opposed to stretched tight and therefore smooth. Vaginal tears, spontaneous as well as during intercourse, perhaps due to pH increasing fragility of the tissue, is another potential source of discomfort. True, there may be adhesions, cysts, scarring, ptosis, and other blockages that may also prevent ovulation, but 60% of conception is mental and comfort is a significant factor. So is orgasm. Without orgasm and the vagina to help move the sperm towards the egg, the odds of conception also diminish, and orgasm is as much mental- and comfort-related as it is physical. Before subjecting patients to hormone therapy to aid in conception, consider helping them to find ways to be more comfortable, especially since hormone therapy will likely and significantly exacerbate P&S dysfunction. Balancing the Oxidant-Antioxidant ratio [66, 67, 68, 69, 70], hormones [48, 71], and the P&S nervous systems may lead to more normal odds of conception, as well as a healthier and more normal pregnancy [1].
To promote conception, a woman must have the proper mindset, including being comfortable and as pain free as possible. Her comfort promotes orgasm. Without orgasm, it seems as if the odds of conception are very low. With orgasm, it seems as if the odds of conception are significantly higher. Orgasm helps to propel sperm to the egg. Therefore, pain could be a significant factor in the appearance of infertility. Lifestyle, urogynecological, and even some disorders such as Ehlers Danlos Syndrome or Hypermobility (EDSh) may involve pain and thereby prevent conception. In the case EDSh, many of these women are essentially born with pain and have never experienced what it feels like to be pain-free. Although their conscious perception of pain may seem minimal, the underlying neural activity at the brainstem level tells a very different story. As a connective tissue disorder, EDSh does not permit a woman to tighten her vagina when excited. This may cause more friction during intercourse and thereby more pain. However, the EDSh female’s pain threshold is high, masking the perception of pain, so no countermeasures for pain are taken and she fails orgasm and thereby fails conception.
While the connective tissue looseness leading to excessive wrinkling is not treatable, reducing the pain may be. Note, most EDS/h women that have undergone fertility treatments for the first or second baby (babies) almost always have a subsequent surprise baby. This proves that they were not infertile to begin with. It may be that the delivery of the first babies stretched her just enough that less wrinkling occurred, enabling orgasm, thereby promoting conception. The woman’s comfort is important for conception.
Clinical Implications

Key Points: PE may interfere with ovulation, implantation, and libido.
Clinical Takeaway: Reducing PE through hydration, sleep hygiene, and stress modulation may improve fertility outcomes.
Action Step: Evaluate P&S patterns before prescribing fertility hormones.
Gestational (Pregnancy & Delivery) Issues, Part 1 - Introduction
Most P&S dysregulation patients are significantly dehydrated. Pregnancy for women with P&S dysregulation tends to help them to feel better. The additional fluid volume they are carrying around the fetus helps them with their overall blood volume and resultant perfusion of the heart and brain and the structures in between. Most of their P&S dysfunction symptoms are relieved. We tease our pregnant patients that they are “cheating,” that during pregnancy, they have an extra pump inside helping them! 😊
Non-cardiovascular complications in pregnancy are relatively rare. They may include: preterm birth, Antepartum Hemorrhage, Postpartum Hemorrhage, Hyperemesis Gravidarum, Shoulder Dystocia, Caesarean wound infection, postpartum Psychosis, Post-Traumatic Stress Disorder, precipitate labor, and being born before arrival at intended places of birth. These may have profound impact on the perinatal and neonatal outcomes and hemodynamic stability, including elevated Heart Rates. The elasticity of soft vessels during pregnancy, due to Progesterone, increases the risk of profound Varicose Veins in the legs and Vulva [72]. Increased joint elasticity and pain, including Pelvic Girdle pain and Pelvic instability, may be seen postdelivery [73]. Analgesics may be less effective than local anesthetics in P&S dysregulation patients. While initially it was thought that a patient with Postural Orthostatic Tachycardia Syndrome (POTS) would benefit from Cesarean delivery [74], the majority of patients with POTS are seemingly able to birth vaginally [75]. However, if pain becomes intolerable at birth, Cesarean section may be indicated [76].
A whole-body, 11-system, Preconception Screening is an option for those who are concerned about a difficult pregnancy to establish a baseline to track changes that may occur during pregnancy. This may include additional testing, including cardiovascular testing, P&S monitoring, Pulmonary Function Testing (if shortness of breath), etc., depending on symptoms. Of course, pregnancy monitoring and delivery planning, ensuring that the hospital has the facilities and personnel who understand P&S dysregulation and its associated disorders (including connective tissue disorders), in case of emergencies or difficulties, is essential. For example, any surgery (e.g., C-Section or Episiotomy) that may be required will require a (General) Surgeon who understands the fragility and laxity of the tissue in connective tissue disorder patients, and they will require suturing of many more layers and many closer stitches, since their tissues do not heal as would a normal patient. This will help to prevent re-stitching and very wide and painful scarring but will not prevent all scarring.
Generally, any pregnancy in which the mother has a diagnosis beforehand that suggests a high-risk pregnancy may only need closer monitoring and less stressful lifestyle during gestation. There are many high-risk pregnancies every year, so being high-risk is not by itself a problem. Profiling a patient’s risk means the patient and baby will benefit from watching the pregnancy a little more closely with their physician, and being aware of possible complications that may arise and knowing how to handle them, including: (1) joint subluxations, (2) circulatory disorders (e.g., blood pooling, “leaky” and “stretchy” blood vessels), (3) breakthrough bleeding and miscarriage, and (4) uterine rupture or premature rupture of membranes. Genetic testing of the fetus may be recommended. High risk deliveries are not a significant concern any longer, especially if the physician is prepared.
Clinical Implications

Key Points: Non-cardiovascular complications in pregnancy are relatively rare.
Clinical Takeaway: Generally, any pregnancy in which the mother has a diagnosis beforehand that suggests a high-risk pregnancy may only need closer monitoring and less stressful lifestyle during gestation.
Action Step: A whole-body, 11 system, Preconception Screening is an option for those who are concerned about a difficult pregnancy to establish a baseline to track changes that may occur during pregnancy.
Gestational (Pregnancy & Delivery) Issues, Part 2 – Oxidative Stress
Normal placental health relies on proper Antioxidant-Oxidant balance. Oxidants (Reactive Oxygen Species) are actually required to stimulate growth and signal stages of development, and antioxidants are then required to protect the fetal tissues that are developed [77, 78]. Unbalanced or mis-timed placental Oxidative Stress may give rise to a variety of complications, including miscarriage [79, 80, 81], recurrent pregnancy loss [82], and preeclampsia, amongst others [83, 84, 85, 86, 87, 88, 89, 90, 91, 92]. Collagen is the “soft skeleton” of the body and supports all structures that have form and function. Extracellular Matrix and Collagen turnover in the early stages of fetal development have been observed to be decreased in patients with early pregnancy loss. Collagen levels negatively correlate with increased Oxidative Stress, possibly accounting for the heightened placental vascular resistance and Endothelial dysfunction secondary to decreased and dysregulated collagen turnover [93]. This highlights the importance of pre-natal vitamins and a well-balanced and healthy diet replete with natural sources of antioxidants, vitamins, minerals, and nutrients. It also highlights the importance of avoiding smoking, drugs, and too much alcohol (a small glass of wine with dinner on occasion may be accepted, but nothing else). Given the placental barrier, many supplements do not cross the placental barrier. This further highlights the need for a healthy and well-balanced diet to receive the necessary nutrients and antioxidants in forms that will cross the placental barrier [94, 95, 96, 97, 98, 99, 100, 101, 102].
Oxidative Stress may complicate pregnancy, possibly leading to spontaneous abortion, recurrent pregnancy loss, Preeclampsia, and pre-term labor. The impact of maternal lifestyle may also effect pregnancy, including extremes of body weight and lifestyle factors such as (1) cigarette smoking, (2) alcohol use, and (3) recreational drug use, which promote Oxidative Stress, as well as (4) excessive exposures to environmental pollutants and (5) excessive consumption of processed foods, both from the processing additives and the removing of nutrients, which could affect not only fertility, but pregnancy as well [1]. [103, 104, 105, 106]
Again, antioxidant supplementation (e.g., r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent) may be effective in controlling Oxidative Stress. Exercise, arguably the most potent of antioxidants, during pregnancy [107, 108, 109, 110] reduces Oxidative Stress as well as having many other benefits, including core strength to help with labor, delivery and recovery.
During normal pregnancy, immune (Leukocyte) activation produces an inflammatory response. This is associated with increased production of “super antioxidants” (Superoxide anions) in the 1st trimester [111, 112]. Importantly, Oxidative Stress during the second trimester of pregnancy is considered a normal occurrence to balance the fetal development progressing and is supported by mitochondrial production of Reactive Oxygen Species and vitamin E in the placenta that increases as gestation progresses [113, 114, 115, 116].
Pregnancy has been observed to help many women that have difficult menarche and early menstruation. One possible reason is that somewhere in the process, the woman’s P&S systems came into balance, which helped to normalize her reproductive hormones, which enabled more normal reproductive health. Another possible reason is that the additional fluid carried during the pregnancy helps to properly hydrate and perfuse the brain and helps to naturally and appropriately increase the stroke volume (essentially the physical size) of the heart, helping the heart to pump more blood throughout the rest of the woman’s life.
Clinical Implications

Key Points: Normal placental health relies on proper Antioxidant-Oxidant balance.
Clinical Takeaway: Oxidants (Reactive Oxygen Species) are actually required to stimulate growth and signal stages of development, and antioxidants are then required to protect the fetal tissues that are developed
Action Step: Establish and maintain a proper oxidant/antioxidant balance.
Gestational (Pregnancy & Delivery) Issues, Part 3 – Preeclampsia
Preeclampsia clinically presents as a blood pressure reading > 140/90 mm Hg, taken on two separate occasions at least 6 hours apart along with proteinuria (≥ 0.3 g protein in a 24-hour urine specimen or persistent 1+ (30 mg/dL) protein on dipstick) after 20 weeks of gestation. It is a complex multisystem disorder that may affect previously normotensive women [117, 118]. The major pathophysiologic disturbances are focal vasospasm and a porous vascular tree [119, 120, 121], which may be a result of lax connective tissue (e.g., including in women with Ehlers-Danlos Syndrome or Hypermobility) in the vascular tree due to excess Progesterone possibly due to P&S dysregulation, including PE. Other possibilities include:
Interactions between vasodilators and vasoconstrictors, such as Nitric Oxide, Endothelin 1, Angiotensin II, Prostacyclin, and Thromboxane
Excessive maternal Oxidative Stress, which may lead to endothelial cell dysfunction [118, 122] and systemic vasoconstriction [123] (normal Oxidative Stress is important for normal physiological functions and for placental development [124]); Preeclampsia, however, represents a much higher state of Oxidative Stress than normal pregnancies do [125])
Excessive placental Oxidative Stress [140-148] associated with elevated levels of protein Carbonyls, Lipid Peroxides, Nitrotyrosine residues, DNA Oxidation, and other Reactive Oxygen Species or free radicals [78,149-171].
Affected women also have decreased antioxidant levels [149,152, 126] and low levels of Vitamins C and E (two antioxidants) [151]. Inadequate Vitamin C intake seems to be associated with an increased risk of Preeclampsia [127] and some studies have shown that peri-conception supplementation with multivitamins may lower the risk of Preeclampsia in normal or under-weight women [128, 129].
Low birth weight may be a function of excess Oxidative Stress [172-180]. Another weight issue is abnormally low or excessively high maternal weight during pregnancy. Either condition may lead to complications during pregnancy (e.g., Preeclampsia) or pre-term labor, and these abnormal weight conditions may also be a function of Oxidative Stress [130] or endothelial dysfunction [131, 132, 133]. The state of labor, whether preterm or term, necessitates the actions of Glutathione Peroxidase to limit lipid oxidation and is associated with a Reactive Oxygen Species-induced reduction of antioxidant defenses [134]. Women with preterm labor as opposed to women with term deliveries have diminished antioxidant abilities to defend against Oxidative Stress-induced damage [135, 136, 137, 138]. Figure 4 is an example of the involvement of the immune system in pregnancy, in this case, implantation. Dendritic cells, Natural Killer cells, Lymphocytes, Monocytes, etc., involve an appropriate oxidant-antioxidant balance for a normal pregnancy.
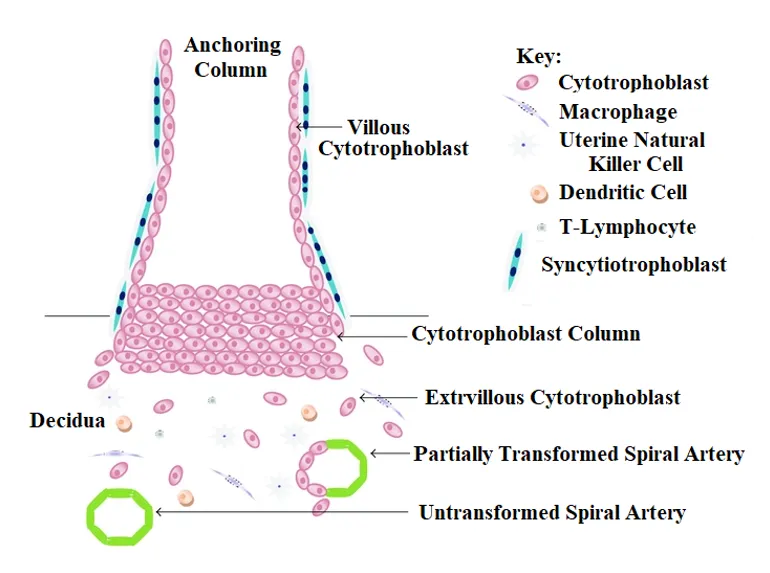
Figure 4: Diagram of the Maternal Fetal Interface in early pregnancy. Extravillous Cytotrophoblast cells migrate away from the cell column of the anchoring villus (the Anchoring Column) and invade through the maternal Deciduas and inner third of the myometrium in order to gain access to maternal blood supply via maternal Spiral Arteries. Transformation of the maternal Spiral Arteries occurs as Endovascular Trophoblast help convert the Endothelial Cells lining the arteries into an amorphous fibrinoid matrix which anchors the fetus to the mother. Oxidative stress is generated during normal placental development. The antioxidants Nitric Oxide, Copper, Zinc, Manganese, and Vitamins C and E in pregnancy, and Selenium via its incorporation into the Glutathione Peroxidase cycle, have been shown to be essential antioxidants, whose activity promotes heathy pregnancy. [139]
Clinical Implications

Key Points: Preeclampsia a complex multisystem disorder that may affect previously normotensive women.
Clinical Takeaway: The major pathophysiologic disturbances are focal vasospasm and a porous vascular tree, possibly due to excess Progesterone. Causes may also include immune and P&S dysregulation during pregnancy leading to high BP and excess Progesterone.
Action Step: Frequent monitoring of blood pressure and for proteinuria throughout pregnancy aids in early detection.
Post-Gestational (Postpartum) Recovery and Wellness
Bringing new life into the world is a remarkable experience, yet the postpartum period is also a time of profound physical and emotional adjustment that deserves compassionate attention and care. Post-gestational or Postpartum issues include common physical changes like hair loss and constipation, as well as more serious complications such as postpartum hemorrhage, pre-eclampsia, severe headaches, fever, dizziness, severe mood swings, depression, abnormal scar formation, and Pelvic Prolapses, which may be associated with Episiotomy, Venous Thrombosis, complicated Perinea wounds and Coccyx dislocation, excessive bleeding, as well as chest pain, blood clots, and infection. Stress-induced Urinary Incontinence may also follow due to weakened Pelvic Floor [181]. All are treatable.
Post-Partum Depression is likely due to the sudden loss of the extra volume that was carried during pregnancy helping to perfuse the brain. The sudden loss of volume may be compounded by varicose veins caused by the pregnancy and tissue laxity, causing more Orthostatic dysfunction (POTS, Orthostatic Intolerance, or Orthostatic Hypotension), a type of P&S dysregulation, leading to the poor cerebral perfusion and depression-like and even anxiety-like symptoms. This may be relieved by proper daily hydration with electrolytes to maintain proper volume and, as needed, vasopressor therapy to relieve the Orthostatic dysfunction. With varicose veins, deep vein thromboses (DVTs) are possible. Sudden onset of Tachycardia and shortness of breath may represent pulmonary embolism due to DVT.
It is common for hair to thin for up to five months after birth as hormone levels shift. Hormone levels may also lead to constipation after childbirth. Note, the significant decrease in Estrogen post-delivery may lead to a decrease in Serotonin levels affecting both bowel motility leading to constipation, as well as behavioral health disorders, including depression. Constipation may be managed with increased fiber and water intake for some patients, especially if they are dehydrated, or Osmotic laxatives: Polyethylene glycol (e.g., MiraLAX) or magnesium citrate or glycinate at bedtime to relax intestinal muscles, or sometimes a stool softener or laxative. Lower GI (bowel) conditions may be a sign of a more whole body dysregulation. To help with overall wellness, consider pre/probiotics and foods to improve gut quality; walking; breathing work; nutrient support, including daily vitamin C to support collagen and peristalsis; B-complex vitamins, especially B6 and B12 for stress modulation and energy; Omega-3s (DHA/EPA) as an anti-inflammatory to improve gut-brain communication.
Progesterone also decreases Serotonin levels which may affect bowel motility leading to constipation. However, Progesterone levels drop sharply after delivery. This rapid decrease is normal, but may contribute to depression or irritability. Progesterone levels may take weeks to months to fully stabilize, depending on whether the person is breastfeeding. The changes in hormone levels are exacerbated by the fact that, upon delivery, the woman also lost the fluid of pregnancy, which may reduce cerebral perfusion and cause a greater cardiac workload to replace. The greater cardiac workload coupled with high blood pressure that may be attempting to compensate for poor coronary and cerebral perfusion is possible and may be prevented or relieved with proper daily hydration with the appropriate electrolytes. Sudden chest pain or a rapid, irregular heartbeat may also be a compensatory mechanism, similar to high BP to attempt to move more blood to the heart and the brain.
If the heart became deconditioned during pregnancy, the heart may not be able to compensate for the greater cardiac workload. This may be further exacerbated by the weight of the pregnancy damaging the veins in the legs, preventing them from helping to move blood to the abdomen to reduce cardiac workload. Damage to the veins may also cause blood clots (DVTs), which may also compromise blood flow to the heart and thereby the brain. Poor cerebral perfusion leads to lightheadedness, fatigue, brain fog, headache/migraine, sleep difficulties, memory and cognitive difficulties, sensory abnormalities (including vision changes or dizziness), in addition to the behavioral health issues, including depression, anxiety, bipolar, postpartum psychosis, and other behavioral health disorders that lead to infant abandonment, delusions and hallucinations, overwhelming feelings of worthlessness and guilt, obsessive-compulsive disorder, all potentially leading to a mother being unable to care for or bond with her baby. Proper daily hydration is important to help maintain proper blood flow to the heart and, especially the brain, to minimize the effects of the loss of fluid and the resulting poor cerebral perfusion.
Clinical Implications

Key Points: Pregnancy naturally rebalances P&S tone and increases perfusion.
Clinical Takeaway: Women who feel better during pregnancy may benefit from mimicking this hemodynamic, hydration, and Autonomic stability postpartum.
Action Step: Support P&S and cardiovascular tone and hydration in postpartum recovery plans.
Pre- and Peri-Menopause and Active Menopause
As decreasing levels of Estrogen compound the age-related decrease in antioxidant production, Oxidative Stress also increases with Menopause [185]. This increases the risk of depression, anxiety, and other behavioral health disorders leading to reduced emotional well-being, mood instability, or diminished quality of life. The effects of Oxidative Stress on the P&S nervous systems also lead to both Sympathetic Withdrawal (SW, an alpha-Adrenergic or alpha-Sympathetic dysregulation underlying Orthostatic Dysfunction) and Parasympathetic Excess (PE).
Persistent PE beyond the childbearing years often results in more symptomatic or challenging menopausal transitions, including prolonged or intense perimenopausal phases. This includes the symptoms of Hot Flashes (Vasomotor Syndrome), Night Sweats, and the psychological disorders typically associated with menopause. The Parasympathetics control the hormones, especially the reproductive hormones in women. Given that the Thyroid is the master gland outside the brain, and the fact that the Parasympathetics help to control the Thyroid, a complete Thyroid panel should be assessed to ensure that is functioning properly. PE may reduce Thyroid hormone release and be a cause for hormone replacement therapy, especially at the higher doses. Changing Thyroid function also changes PE, and both should be stabilized before attempting to balance the reproductive hormones. With proper Thyroid function, the remaining reproductive hormone imbalance and associated P&S imbalance may be addressed.
PE was initially discovered in women who have had more than two pregnancies. In these cases, pregnancy is defined as a pregnancy that lasts at least through the first trimester before termination. Symptoms of PE are those attributed to motherhood; therefore, it was originally going to be labelled “Motherhood Syndrome.” However, we started to find it in men and couldn't call it that anymore.
PE seems to lead to women entering Menopause early or causing them to have miserable Menopauses, including more frequent and serious night sweats and behavioral health symptoms and more frequent Hot Flashes (Vasomotor Syndrome). Relieving PE before Menopause helps normalize the timing of its onset and normalize the frequency and severity of the symptoms, and in some cases eliminate at least the night sweats and behavioral health symptoms.
Oxidative Stress continues to effect Women’s Health into Menopause. Oocyte quality decreases in relation to increasing maternal age apparently due to mitochondrial changes possibly caused by excessive Reactive Oxygen Species production (Oxidative Stress) [182]. Oxidative stress, iron stores, blood lipids, and body fat typically increase with age, especially after Menopause. The cessation of Menses leads to an increase in iron levels throughout the body. Elevated iron stores could induce oxidative imbalance [183]. Menopause also leads to a decrease in Estrogen and the loss of its protective effects against oxidative damage to the Endometrium [184]. This may be compounded by the negative effect of Oxidative Stress on the P&S nervous systems and their reduced or less efficient control and coordination of the Hormone system. Hormone Replacement Therapy may be beneficial against Oxidative Stress by antagonizing the effects of lower Antioxidant levels that normally occurs with aging. Of course, supplementing natural Antioxidant levels (e.g., r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent) is also beneficial against Oxidative Stress and helps to preserve or re-energize the nervous system.
It is well known that reproductive hormones in certain parts of the nervous system act as neurotransmitters. This highlights the intimacy between the two systems. From the P&S systems, Vasomotor Syndrome and Sweating are functions of Sympathetic activity. With PE, Sympathetic activity is amplified and the resulting vascular and sweat gland control is exaggerated, leading to the Hot Flashes and Night Sweats. The PE itself leads to the psychological disorders, including depression (due to PE) and anxiety (due to secondary Sympathetic Excess (SE), when stressed). Relieving PE and thereby secondary SE often prevents these symptoms or relieves these symptoms if they've already presented.
In addition to PE, Oxidative Stress is associated with depression, anxiety, and mood instability. This may be associated with Estrogen depletion causing a lower Superoxide Dismutase level which is associated with higher depression and anxiety scores in postmenopausal women [185]. Ovarian aging leads to unpredictable production of estrogen unless it reaches a permanent low level, which causes various vasomotor symptoms which interfere with sleep leading to anxiety and depression that are highly prevalent due to hormonal changes [186, 187]. Indigenous antioxidant levels deteriorate with decreased levels of estrogen which reinforces risk of oxidative stress-related diseases in menopause [188, 189, 190, 191]. Postmenopausal women who may be susceptible to developing Oxidative Stress should avail the benefits of a diet which is rich in antioxidants so that their psychological disturbances may be improved and effects of oxidative damage may be reduced [192, 193].
Clinical Implications

Key Points: Autonomic dysregulation drives hot flashes, sleep disturbance, and mood instability.
Clinical Takeaway: Addressing both sympathetic and parasympathetic imbalances (breathing therapy, HRV biofeedback, adaptogens) can reduce symptom burden.
Action Step: Screen menopausal patients for autonomic symptoms before adding pharmacologic therapy.
Histamine: Menopause, SIBO, and Low Thyroid Levels
As stated throughout, there is a very remarkable connection between Histamine and Women’s Health. In Menopause, women are more likely to develop Histamine Intolerance. This is because both Estrogen and Progesterone drop during Menopause. For many women, Progesterone ends up even lower than Estrogen. So, they may remain Estrogen-dominant while in Menopause.
Estrogen is the connection to Histamine. Estrogen reduces an important histamine-degrading enzyme, Diamine Oxidase. Diamine Oxidase is very vulnerable to gut infections like SIBO. Gut infections destroy the body’s ability to make Diamine Oxidase. So SIBO and Estrogen dominance together significantly elevate Histamine levels, leading to many other symptoms, including unexplained food and chemical allergies, Asthma-like symptoms, abnormal periods, unexplained rashes and hives, and in the extreme, anaphylaxis.
In theory, balancing Hormones helps. However, synthetic hormones often make Histamine intolerance worse. Synthetic Hormone replacement is clearly linked to the onset of allergies and asthma. Especially in patients with MCAS, the fillers and capsules as well as the synthetic Hormones themselves often cause more Mast Cell activation and thereby Histamine release. Even within the synthetic Hormone options, this maybe the difference between tolerating name brand versus generic (e.g.. Synthroid® vs. a generic). Bioidentical hormones may work for some women.
Low thyroid levels may also contribute to Histamine excesses [194]. This is likely due to reductions in Thyroid hormones that help regulate Mast Cells and reduce Histamine production. If levels of Thyroid hormone are reduced, Histamine levels may be much higher. It may also be due to Parasympathetic Excess (PE) which is known to be associated with Hypothyroidism and also the excessive release of Mast Cells, together with causing Sympathetic Excess (SE) which releases too much Histamine from each Mast Cells, further amplifying the excessive response.
Clinical Implications

Key Points: Low thyroid levels may also contribute to Histamine excesses.
Clinical Takeaway: PE causes SE which releases too much Histamine from each Mast Cells, further amplifying the excessive response.
Action Step: Normalizing PE and Thyroid function helps to relieve Menopause-related symptoms.
Summary: Integrating the P&S System, Oxidative Stress, and Women’s Health
Women’s health across the life span – from menarche to menopause – is governed by the intricate balance between the Parasympathetic (P) and Sympathetic (S) branches of the Autonomic Nervous System. These two systems form the body’s regulatory foundation, orchestrating hormonal, immune, and metabolic responses that directly affect fertility, menstruation, pregnancy, and menopausal transitions.
The Parasympathetic system (“rest, repair, restore, protect”) uniquely coordinates hormonal, immune, and gastrointestinal functions, while the Sympathetic system (“fight or flight”) modulates stress, circulation, and energy mobilization. Dysregulation in this balance, manifesting as Parasympathetic Excess (PE) or Sympathetic Excess (SE), is a common underlying factor in many female conditions. PE, in particular, has been linked to early or symptomatic menopause, menstrual irregularities, post-gestational changes, and disorders such as PCOS, Endometriosis, and Mast Cell activation
Oxidative Stress acts as a compounding mechanism throughout the female life cycle. Elevated reactive oxygen and nitrogen species impair oocyte quality, promote inflammation, and contribute to uterine and ovarian dysfunction. Conditions like infertility, recurrent pregnancy loss, and preeclampsia share oxidative stress as a biological hallmark. The decline in Estrogen during perimenopause further intensifies oxidative imbalance, exacerbating symptoms such as mood instability, anxiety, vasomotor changes, and poor tissue resilience
Moreover, Mast Cell activation – under Parasympathetic control – links immune hyper-reactivity with hormonal and neurovascular symptoms. The P&S imbalance magnifies this cascade, producing systemic manifestations ranging from dysmenorrhea and fibroids to fatigue and cognitive fog.
Clinically, restoring autonomic balance through lifestyle modification, nutrition, hydration, and targeted antioxidant therapy (e.g., r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent) offers a pathway to recovery. Stabilizing thyroid and reproductive hormone feedback loops, optimizing stress resilience, and addressing oxidative stress help normalize cycle regularity, fertility, and menopausal quality of life.
In essence, women’s health cannot be compartmentalized by organ or age. The P&S nervous systems provide a unifying lens – linking the brain, hormones, and body into one regulatory continuum. Understanding and supporting the P&S system is therefore foundational to achieving long-term vitality, emotional stability, and women’s well-being.
Women are not just small men!* Given that the P&S systems help to regulate a woman’s entire lifecycle, from Menarche through Menopause, they provide more information to help differentiate women’s physiology from men’s. In general, women are still going to need HRT, but they may not need high doses. For example, if patients go on HRT and they start bleeding worse, and the doctor says there's nothing more that may be done, P&S therapy may provide them with an alternative. P&S medicine may involve other provider types to monitor, diagnose, and treat, such as Primary Care Providers, Concierge doctors, GPs, and Integrative and Functional Medicine doctors. Eventually, OB-GYNs will come around, if not already, as they start losing patients, because somebody else has figured out how to normalize women’s physiology. P&S monitoring helps to provide therapy which may act as a curative as well as a preventative.
Next Steps
To develop protocols (treatments and therapies) that move from hormone replacement therapy as the end-all and be-all, to autonomic therapy, perhaps mast cell therapy, and perhaps other things. This includes domestically and reliably sources rALA. Two agents, two plant-based medicine supplements, for women's health may be to relieve (1) “Hot Flashes” (Vasomotor Syndrome) and (2) Pelvic Floor issues. The second one would help with some men as well, but the whole Pelvic Floor. Night Sweats and Behavioral Health issues are at least in part relieved by relieving PE, but not Hot Flashes, the third in the common quality of life concerns for menopausal women. P&S Monitoring will provide more information for these protocols to be both preventative and curative through women’s life cycles.
This is just the start. Just like we're building on the fact that patients are going to be voting with their feet, thereby moving medicine forward in the next several years, women have taken over medicine. Women have finally decided to differentiate themselves and how they are managed from men. This seems to be the ground floor. With P&S Monitoring providing foundational and causal information, including controlling Hormones and Mast Cells, now is the time to integrate the additional information from P&S Monitoring into better manage of women; both women in general and women as doctors managing themselves.
While the Ob/Gyn is going to have healthier patients, the perception is that they are going to lose as hormone replacement therapies are reduced. However, they will not be eliminated. Women are still going to want/need birth control (which is hormone replacement therapy) and hormone replacement therapy in later years during menopause. The true benefit to an Ob/Gyn is more information to determine why many of the women that they treat with hormones are not relieved, and, if anything, are made worse.
Another benefit is that most women for whom hormone replacement therapy does not help, are dismissed or marginalized. The alternative to hormone replacement therapy for those in whom it does not work is to help retain those patients by providing viable alternative(s). Furthermore, these alternatives to hormone replacement therapy will also affect other symptoms, and now the Gynecologist provides additional reasons for retaining patients. For example, COVID and the current respiratory viruses. Gynecologists that are using P&S Monitoring will have additional information to also treat these other conditions, almost acting as a Primary Care Physician, helping to increase their practice. Reversing the current trend of patients using their PCP as their Gynecologists as well.
As another example, a PCP we work with came highly recommended because the patient’s Gynecologist seems to be ignoring all her pre-menopausal symptoms and just telling her that she's got to go through the process. The patient refused to accept that as an answer, and she needs somebody else to give her more in-depth information. She found this PC that had more information from P&S Monitoring and found relief. Many more women are going to Concierge doctors, for example, because they are not making any progress with a regular doctor, particularly in women's health
Glossary of Core Mechanisms adverse to Women’s Health with Treatment and Therapy Options
Parasympathetic Excess (PE)
An over-activation of the Parasympathetic nervous system leading to fatigue, hypotension, vasodilation, dizziness, heavy menses, headache or migraine, or depression, and other behavioral health symptoms and more due to poor cerebral perfusion.
Treat with (1) very low-dose anti-cholinergics, e.g., Nortriptyline, nor more than 10.0 mg, qd(7PM), or (2) Low-and-Slow exercise, up to 40 minutes/day for six to nine months
Sympathetic Excess (SE)
An over-activation of the beta-Sympathetic nervous system producing tension, elevated heart rate or blood pressure, sleep difficulties, inflammation, and anxiety, and other behavioral health symptoms and more while attempting to compensate for poor cerebral perfusion.
Treat gently, if quality of life is disturbed while PE & SW are being relieved, with low-dose sympatholytics, history dependent (e.g., if high heart rate disturbs quality of life, Propranolol, 10 mg, tid, prn).
Sympathetic Withdrawal (SW)
An insufficient activation of the alpha-Sympathetic nervous system producing Orthostatic dysfunction, contributing to poor cerebral perfusion and more.
Treat with (1) Midodrine, 2.5 mg, tid, titrated from 1.25 mg, qdAM, in six steps over 12 weeks, or (2) r-Alpha-Lipoic Acid, 600 mg, tid, or equivalent.
Oxidative Stress (OS)
A biochemical imbalance between free radicals and antioxidants, contributing to cellular injury, hormonal disruption, and accelerated aging.
Mast Cell Activation Syndrome (MCAS)
A hypersensitive immune state where Mast Cells release excessive histamine and inflammatory mediators, often triggered by stress, estrogen fluctuations, or dysautonomia.
Treat with both (are required): H1 blocker Zyrtec 10 mg, qd and H2 blocker Pepcid, 20 mg qd.
P&S Dysregulation
A condition in which Parasympathetic and Sympathetic functions fail to maintain equilibrium, leading to disrupted metabolism, hormonal imbalance, immune dysregulation, gastrointestinal dysmotility, and inflammatory responses across the female lifecycle.
Frequently Asked Questions
What should I expect from life coaching?
Life coaching helps you clarify goals, overcome challenges, and create actionable plans for personal and professional growth in a supportive, goal-focused environment.
How long before I see results?
Results vary, but many clients notice positive changes after a few sessions with consistent effort and commitment.
Is life coaching for both personal and career goals?
Yes! Our coaching supports both personal development and career growth, helping you achieve success in all areas of life.
Do I need to prepare for my session?
No special preparation is needed, but coming with an open mind and clear goals will help maximize your coaching experience.
REFERENCES
* Quoted from the philosophy coined by Dr. Stacy Sims
[1] Agarwal A, Aponte-Mellado A, Premkumar BJ, Shaman A, Gupta S. The effects of oxidative stress on female reproduction: a review. Reprod Biol Endocrinol. 2012 Jun 29;10:49. doi: 10.1186/1477-7827-10-49. PMID: 22748101; PMCID: PMC3527168. Figure: https://www.ncbi.nlm.nih.gov/core/lw/2.0/html/tileshop_pmc/tileshop_pmc_inline.html?title=Click%20on%20image%20to%20zoom&p=PMC3&id=3527168_1477-7827-10-49-1.jpg Accessed 19Jul23.
[2] Binda MM, Donnez J, Dolmans MM. Targeting mast cells: a new way to treat endometriosis. Expert Opin Ther Targets. 2017 Jan;21(1):67-75. doi: 10.1080/14728222.2017.1260548. Epub 2016 Nov 28. PMID: 27841046.
[3] Al-Gubory KH, Fowler PA, Garrel C. The roles of cellular reactive oxygen species, oxidative stress and antioxidants in pregnancy outcomes. Int J Biochem Cell Biol. 2010 Oct;42(10):1634-50. doi: 10.1016/j.biocel.2010.06.001. Epub 2010 Jun 19. PMID: 20601089.
[4] Fisher-Wellman K, Bell HK, Bloomer RJ. Oxidative stress and antioxidant defense mechanisms linked to exercise during cardiopulmonary and metabolic disorders. Oxid Med Cell Longev. 2009;2:43–51.
[5] Fisher-Wellman K, Bell HK, Bloomer RJ. Oxidative stress and antioxidant defense mechanisms linked to exercise during cardiopulmonary and metabolic disorders. Oxid Med Cell Longev. 2009;2:43–51.
[6] Fisher-Wellman K, Bloomer RJ. Acute exercise and oxidative stress: a 30 year history. Dyn Med. 2009;8:1.
[7] Leelarungrayub D, Saidee K, Pothongsunun P, Pratanaphon S, YanKai A, Bloomer RJ. Six weeks of aerobic dance exercise improves blood oxidative stress status and increases interleukin-2 in previously sedentary women. J Bodyw Mov Ther. 2011;15:355–362.
[8] Chandra A, Surti N, Kesavan S, Agarwal A. Significance of oxidative stress in human reproduction. Arch Med. 2009;5:528–542.
[9] van den Berge M, Heijink HI, van Oosterhout AJ, Postma DS. The role of female sex hormones in the development and severity of allergic and non-allergic asthma. Clin Exp Allergy. 2009 Oct;39(10):1477-81. doi: 10.1111/j.1365-2222.2009.03354.x. PMID: 19954427.
[10] Dempsey T, Rosenthal L. Chapter 24. Urogynecology & Hypermobility. In: Disjointed Navigating the Diagnosis and Management of Hypermobile Ehlers-Danlos Syndrome and Hypermobility Spectrum Disorders. Jovin D. (ed.). Hidden Stripes Publications, Inc. 2020.
[11] Heitz NA, Eisenman PA, Beck CL, Walker JA. Hormonal changes throughout the menstrual cycle and increased anterior cruciate ligament laxity in females. J Athl Train. 1999 Apr;34(2):144-9. PMID: 16558557; PMCID: PMC1322903.
[12] Giudice LC, Kao LC. Endometriosis. Lancet. 2004 Nov 13-19;364(9447):1789-99. doi: 10.1016/S0140-6736(04)17403-5. PMID: 15541453.
[13] Mier-Cabrera J, Jiménez-Zamudio L, García-Latorre E, Cruz-Orozco O, Hernández-Guerrero C. Quantitative and qualitative peritoneal immune profiles, T-cell apoptosis and oxidative stress-associated characteristics in women with minimal and mild endometriosis. BJOG. 2011 Jan;118(1):6-16. doi: 10.1111/j.1471-0528.2010.02777.x. Epub 2010 Nov 18. PMID: 21083865.
[14] Rong R, Ramachandran S, Santanam N, Murphy AA, Parthasarathy S. Induction of monocyte chemotactic protein-1 in peritoneal mesothelial and endometrial cells by oxidized low-density lipoprotein and peritoneal fluid from women with endometriosis. Fertil Steril. 2002;78:843–848.
[15] Mier-Cabrera J, Jiménez-Zamudio L, García-Latorre E, Cruz-Orozco O, Hernández-Guerrero C. Quantitative and qualitative peritoneal immune profiles, T-cell apoptosis and oxidative stress-associated characteristics in women with minimal and mild endometriosis. BJOG. 2011 Jan;118(1):6-16. doi: 10.1111/j.1471-0528.2010.02777.x. Epub 2010 Nov 18. PMID: 21083865.
[16] Proctor CJ, Lorimer IA. Modelling the role of the Hsp70/Hsp90 system in the maintenance of protein homeostasis. PLoS One. 2011;6(7):e22038. doi: 10.1371/journal.pone.0022038. Epub 2011 Jul 14. PMID: 21779370; PMCID: PMC3137010.
[17] Mier-Cabrera J, Jiménez-Zamudio L, García-Latorre E, Cruz-Orozco O, Hernández-Guerrero C. Quantitative and qualitative peritoneal immune profiles, T-cell apoptosis and oxidative stress-associated characteristics in women with minimal and mild endometriosis. BJOG. 2011 Jan;118(1):6-16. doi: 10.1111/j.1471-0528.2010.02777.x. Epub 2010 Nov 18. PMID: 21083865.
[18] Arya R, Mallik M, Lakhotia SC. Heat shock genes - integrating cell survival and death. J Biosci. 2007 Apr;32(3):595-610. doi: 10.1007/s12038-007-0059-3. PMID: 17536179.
[19] Beere HM. Death versus survival: functional interaction between the apoptotic and stress-inducible heat shock protein pathways. J Clin Invest. 2005 Oct;115(10):2633-9. doi: 10.1172/JCI26471. PMID: 16200196; PMCID: PMC1236700.
[20] Ciocca DR, Calderwood SK. Heat shock proteins in cancer: diagnostic, prognostic, predictive, and treatment implications. Cell Stress Chaperones. 2005 Summer;10(2):86-103. doi: 10.1379/csc-99r.1. PMID: 16038406; PMCID: PMC1176476.
[21] Rérole A, Gaëtan J, Garrido C. In: Molecular Chaperones: Methods and Protocols. Calderwood SK, Prince TL, editor. Heidelberg: Springer Science+Business Media, LLC; 2011. Hsp70: Anti-apoptotic and tumorigenic protein; pp. 205
[22] Noonan EJ, Place RF, Giardina C, Hightower LE. Hsp70B' regulation and function. Cell Stress Chaperones. 2007 Winter;12(4):393-402. doi: 10.1379/csc-278e.1. PMID: 18229458; PMCID: PMC2134801.
[23] Lambrinoudaki IV, Augoulea A, Christodoulakos GE, Economou EV, Kaparos G, Kontoravdis A, Papadias C, Creatsas G. Measurable serum markers of oxidative stress response in women with endometriosis. Fertil Steril. 2009 Jan;91(1):46-50. doi: 10.1016/j.fertnstert.2007.11.021. Epub 2008 Jan 18. PMID: 18206876.
[24] Chehna-Patel N, Warty N, Sachdeva G, Khole V. Proteolytic tailoring of the heat shock protein 70 and its implications in the pathogenesis of endometriosis. Fertil Steril. 2011 Apr;95(5):1560-7.e1-3. doi: 10.1016/j.fertnstert.2011.01.122. Epub 2011 Feb 21. PMID: 21333981.
[25] Santanam N, Murphy AA, Parthasarathy S. Macrophages, oxidation, and endometriosis. Ann N Y Acad Sci. 2002 Mar;955:183-98; discussion 19-200, 396-406. doi: 10.1111/j.1749-6632.2002.tb02779.x. PMID: 11949947.
[26] Santanam N, Murphy AA, Parthasarathy S. Macrophages, oxidation, and endometriosis. Ann N Y Acad Sci. 2002 Mar;955:183-98; discussion 19-200, 396-406. doi: 10.1111/j.1749-6632.2002.tb02779.x. PMID: 11949947.
[27] Foyouzi N, Berkkanoglu M, Arici A, Kwintkiewicz J, Izquierdo D, Duleba AJ. Effects of oxidants and antioxidants on proliferation of endometrial stromal cells. Fertil Steril. 2004 Oct;82 Suppl 3:1019-22. doi: 10.1016/j.fertnstert.2004.02.133. PMID: 15474067.
[28] Li YQ, Zhang ZX, Xu YJ, Ni W, Chen SX, Yang Z, Ma D. N-Acetyl-L-cysteine and pyrrolidine dithiocarbamate inhibited nuclear factor-kappaB activation in alveolar macrophages by different mechanisms. Acta Pharmacol Sin. 2006 Mar;27(3):339-46. doi: 10.1111/j.1745-7254.2006.00264.x. PMID: 16490171.
[29] Yamaguchi K, Mandai M, Toyokuni S, Hamanishi J, Higuchi T, Takakura K, Fujii S. Contents of endometriotic cysts, especially the high concentration of free iron, are a possible cause of carcinogenesis in the cysts through the iron-induced persistent oxidative stress. Clin Cancer Res. 2008 Jan 1;14(1):32-40. doi: 10.1158/1078-0432.CCR-07-1614. PMID: 18172249.
[30] Van Langendonckt A, Casanas-Roux F, Donnez J. Oxidative stress and peritoneal endometriosis. Fertil Steril. 2002 May;77(5):861-70. doi: 10.1016/s0015-0282(02)02959-x. PMID: 12009336.
[31] Byrne JA, Grieve DJ, Cave AC, Shah AM. Oxidative stress and heart failure. Arch Mal Coeur Vaiss. 2003 Mar;96(3):214-21. PMID: 12722552.
[32] Osborn BH, Haney AF, Misukonis MA, Weinberg JB. Inducible nitric oxide synthase expression by peritoneal macrophages in endometriosis-associated infertility. Fertil Steril. 2002 Jan;77(1):46-51. doi: 10.1016/s0015-0282(01)02940-5. PMID: 11779590.
[33] Kobayashi H, Yamada Y, Kanayama S, Furukawa N, Noguchi T, Haruta S, Yoshida S, Sakata M, Sado T, Oi H. The role of iron in the pathogenesis of endometriosis. Gynecol Endocrinol. 2009 Jan;25(1):39-52. doi: 10.1080/09513590802366204. PMID: 19165662.
[34] Augoulea A, Mastorakos G, Lambrinoudaki I, Christodoulakos G, Creatsas G. The role of the oxidative-stress in the endometriosis-related infertility. Gynecol Endocrinol. 2009 Feb;25(2):75-81. doi: 10.1080/09513590802485012. PMID: 19253102.
[35] Burney RO, Talbi S, Hamilton AE, Vo KC, Nyegaard M, Nezhat CR, Lessey BA, Giudice LC. Gene expression analysis of endometrium reveals progesterone resistance and candidate susceptibility genes in women with endometriosis. Endocrinology. 2007 Aug;148(8):3814-26. doi: 10.1210/en.2006-1692. Epub 2007 May 17. PMID: 17510236.
[36] Parazzini F, Chiaffarino F, Surace M, Chatenoud L, Cipriani S, Chiantera V, Benzi G, Fedele L. Selected food intake and risk of endometriosis. Hum Reprod. 2004;19:1755–1759.
[37] Dragsted LO, Pedersen A, Hermetter A, Basu S, Hansen M, Haren GR, Kall M, Breinholt V, Castenmiller JJ, Stagsted J. et al. The 6-a-day study: effects of fruit and vegetables on markers of oxidative stress and antioxidative defense in healthy nonsmokers. Am J Clin Nutr. 2004;79:1060–1072.
[38] Huang HY, Appel LJ, Croft KD, Miller ER 3rd, Mori TA, Puddey IB. Effects of vitamin C and vitamin E on in vivo lipid peroxidation: results of a randomized controlled trial. Am J Clin Nutr. 2002;76:549–555.
[39] Upritchard JE, Schuurman CR, Wiersma A, Tijburg LB, Coolen SA, Rijken PJ, Wiseman SA. Spread supplemented with moderate doses of vitamin E and carotenoids reduces lipid peroxidation in healthy, nonsmoking adults. Am J Clin Nutr. 2003;78:985–992.
[40] Sesti F, Capozzolo T, Pietropolli A, Collalti M, Bollea MR, Piccione E. Dietary therapy: a new strategy for management of chronic pelvic pain. Nutr Res Rev. 2011;25:1–8.
[41] Mier-Cabrera J, Aburto-Soto T, Burrola-Mendez S, Jimenez-Zamudio L, Tolentino M, Casanueva E, Hernandez-Guerrero C. Women with endometriosis improved their peripheral antioxidant markers after the application of a high antioxidant diet. Reprod Biol Endocrinol. 2009;5:1–11.
[42] Mier-Cabrera J, Genera-Garcia M, De La Jara-Diaz J, Perichart-Perera O, Vadillo-Ortega F, Hernandez-Guerrero C. Effect of vitamins C and E supplementation on peripheral oxidative stress markers and pregnancy rate in women with endometriosis. Int J Gynaecol Obstet. 2008;100:252–256.
[43] Hernández Guerrero CA, Bujalil Montenegro L, de la Jara Díaz J, Mier Cabrera J, Bouchán Valencia P. Endometriosis and deficient intake of antioxidants molecules related to peripheral and peritoneal oxidative stress. Ginecol Obstet Mex. 2006;74:20–28.
[44] Paul S, Sharma AV, Mahapatra PD, Bhattacharya P, Reiter RJ, Swarnakar S. Role of melatonin in regulating matrix metalloproteinase-9 via tissue inhibitors of metalloproteinase-1 during protection against endometriosis. J Pineal Res. 2008;44:439–449.
[45] Iwabuchi T, Yoshimoto C, Shigetomi H, Kobayashi H. Oxidative Stress and Antioxidant Defense in Endometriosis and Its Malignant Transformation. Oxid Med Cell Longev. 2015;2015:848595. doi: 10.1155/2015/848595. Epub 2015 Jun 21. PMID: 26185594; PMCID: PMC4491397. Figure: https://static.hindawi.com/articles/omcl/volume-2015/848595/figures/848595.fig.001.jpg. Accessed and modified 19Jul23.
[46] Reis FM, Coutinho LM, Vannuccini S, Luisi S, Petraglia F. Is Stress a Cause or a Consequence of Endometriosis? Reprod Sci. 2020 Jan;27(1):39-45. doi: 10.1007/s43032-019-00053-0. Epub 2020 Jan 6. PMID: 32046437.
[47] Qiu Y, Yuan S, Wang H. Vitamin D status in endometriosis: a systematic review and meta-analysis. Arch Gynecol Obstet. 2020 Jul;302(1):141-152. doi: 10.1007/s00404-020-05576-5. Epub 2020 May 19. PMID: 32430755.
[48] Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004 Jan;81(1):19-25. doi: 10.1016/j.fertnstert.2003.10.004. PMID: 14711538.
[50] Setji TL, Holland ND, Sanders LL, Pereira KC, Diehl AM, Brown AJ. Nonalcoholic steatohepatitis and nonalcoholic Fatty liver disease in young women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2006 May;91(5):1741-7. doi: 10.1210/jc.2005-2774. Epub 2006 Feb 21. PMID: 16492691.
[51] Creanga AA, Bradley HM, McCormick C, Witkop CT. Use of metformin in polycystic ovary syndrome: a meta-analysis. Obstet Gynecol. 2008 Apr;111(4):959-68. doi: 10.1097/AOG.0b013e31816a4ed4. PMID: 18378757.
[52] Palacio JR, Iborra A, Ulcova-Gallova Z, Badia R, Martinez P. The presence of antibodies to oxidative modified proteins in serum from polycystic ovary syndrome patients. Clin Exp Immunol. 2006;144:217–222.
[53] Victor VM, Rocha M, Banuls C, Alvarez A, de Pablo C, Sanchez-Serrano M, Gomez M, Hernandez-Mijares A. Induction of oxidative stress and human leukocyte/endothelial cell interactions in polycystic ovary syndrome patients with insulin resistance. J Clin Endocrinol Metab. 2011;96:3115–3122.
[54] Gonzalez F, Rote NS, Minium J, Kirwan JP. Reactive oxygen species-induced oxidative stress in the development of insulin resistance and hyperandrogenism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2006;91:336–340.
[55] Costello MF, Shrestha B, Eden J, Johnson NP, Sjoblom P. Metformin versus oral contraceptive pill in polycystic ovary syndrome: a Cochrane review. Hum Reprod. 2007;22:1200–1209.
[56] Shai I, Schwarzfuchs D, Henkin Y, Shahar DR, Witkow S, Greenberg I, Golan R, Fraser D, Bolotin A, Vardi H. et al. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N Engl J Med. 2008;359:229–241.
[57] Jose, Antony; Maroli, Roshan; George, Peter1. Autonomic Functions in Hypothyroidism. J Indian Coll Cardiol. Jan–Mar 2023; 13(1):p 23-28. DOI: 10.4103/jicc.jicc_15_22
[58] Al-Gubory KH, Fowler PA, Garrel C. The roles of cellular reactive oxygen species, oxidative stress and antioxidants in pregnancy outcomes. Int J Biochem Cell Biol. 2010 Oct;42(10):1634-50. doi: 10.1016/j.biocel.2010.06.001. Epub 2010 Jun 19. PMID: 20601089.
[59] Fisher-Wellman K, Bell HK, Bloomer RJ. Oxidative stress and antioxidant defense mechanisms linked to exercise during cardiopulmonary and metabolic disorders. Oxid Med Cell Longev. 2009;2:43–51.
[60] Fisher-Wellman K, Bell HK, Bloomer RJ. Oxidative stress and antioxidant defense mechanisms linked to exercise during cardiopulmonary and metabolic disorders. Oxid Med Cell Longev. 2009;2:43–51.
[61] Fisher-Wellman K, Bloomer RJ. Acute exercise and oxidative stress: a 30 year history. Dyn Med. 2009;8:1.
[62] Leelarungrayub D, Saidee K, Pothongsunun P, Pratanaphon S, YanKai A, Bloomer RJ. Six weeks of aerobic dance exercise improves blood oxidative stress status and increases interleukin-2 in previously sedentary women. J Bodyw Mov Ther. 2011;15:355–362.
[63] Burton GJ, Jauniaux E. Oxidative stress. Best Pract Res Clin Obstet Gynaecol. 2011 Jun;25(3):287-99. doi: 10.1016/j.bpobgyn.2010.10.016. Epub 2010 Dec 3. PMID: 21130690; PMCID: PMC3101336.
[64] Ruder EH, Hartman TJ, Goldman MB. Impact of oxidative stress on female fertility. Curr Opin Obstet Gynecol. 2009 Jun;21(3):219-22. doi: 10.1097/gco.0b013e32832924ba. PMID: 19469044; PMCID: PMC2749720.
[65] Chandra A, Surti N, Kesavan S, Agarwal A. Significance of oxidative stress in human reproduction. Arch Med. 2009;5:528–542.
[66] Wang Y, Sharma RK, Falcone T, Goldberg J, Agarwal A. Importance of reactive oxygen species in the peritoneal fluid of women with endometriosis or idiopathic infertility. Fertil Steril. 1997;68:826–830.
[67] Polak G, Koziol-Montewka M, Tarkowski R, Kotarski J. Peritoneal fluid and plasma 4-hydroxynonenal and malonyldialdehyde concentrations in infertile women. Ginekol Pol. 2001;72:1316–1320.
[[68] Polak G, Rola R, Gogacz M, Koziol-Montewka M, Kotarski J. Malonyldialdehyde and anti-oxidant levels in the peritoneal fluid of infertile women. Ginekol Pol. 1999;70:135–140.
[69] Gupta S, Surti N, Metterle L, Chandra A, Agarwal A. Antioxidants and female reproductive pathologies. Arch Med. 2009;5:151–173.
[70] Badaiwy A, Baker El Nashar A, El Totongy M. Clomiphene citrate plus N-acetyl cysteine versus clomiphene citrate for augmenting ovulation in the management of unexplained infertility: a randomized double-blind controlled trial. Fertil Steril. 2006;86:647–650.
[71] Herrero-Mercado M, Waliszewski SM, Caba M, Martínez-Valenzuela C, Gómez Arroyo S, Villalobos Pietrini R, Cantú Martínez PC, Hernández-Chalate F. Organochlorine pesticide gradient levels among maternal adipose tissue, maternal blood serum and umbilical blood serum. Bull Environ Contam Toxicol. 2011 Mar;86(3):289-93. doi: 10.1007/s00128-011-0204-4. Epub 2011 Feb 3. PMID: 21290101.
[72] Pearce G, Bell L, Pezaro S, Reinhold E. Childbearing with Hypermobile Ehlers-Danlos Syndrome and Hypermobility Spectrum Disorders: A Large International Survey of Outcomes and Complications. Int J Environ Res Public Health. 2023 Oct 21;20(20):6957. doi: 10.3390/ijerph20206957. PMID: 37887695; PMCID: PMC10606623.
[73] Pezaro, S., Pearce, G. & Reinhold, E, 'Hypermobile Ehlers‐Danlos Syndrome during Pregnancy, Birth and Beyond: A Review of Midwifery Care Considerations.’ British Journal of Midwifery, 2017; 26(4). DOI: 10.12968/bjom.2018.26.4.217
[74] Glatter KA, Tuteja D, Chiamvimonvat N, Hamdan M, Park JK. Pregnancy in postural orthostatic tachycardia syndrome. Pacing Clin Electrophysiol. 2005 Jun;28(6):591-3. doi: 10.1111/j.1540-8159.2005.50026.x. PMID: 15955196.
[75] Kanjwal K, Karabin B, Kanjwal Y, Grubb BP. Outcomes of pregnancy in patients with preexisting postural tachycardia syndrome. Pacing Clin Electrophysiol. 2009 Aug;32(8):1000-3. doi: 10.1111/j.1540-8159.2009.02430.x. PMID: 19659618
[76] Dutta I, Wilson H, Oteri O. Pregnancy and delivery in ehlers-danlos syndrome (hypermobility type): review of the literature. Obstet Gynecol Int. 2011;2011:306413. doi: 10.1155/2011/306413. Epub 2011 Jun 15. PMID: 21765833; PMCID: PMC3134995.
[77] Lunghi L, Ferretti ME, Medici S, Biondi C, Vesce F. Control of human trophoblast function. Reprod Biol Endocrinol. 2007;5:6.
[78] Nakamura M, Sekizawa A, Purwosunu Y, Okazaki S, Farina A, Wibowo N, Shimizu H, Okai T. Cellular mRNA expressions of anti-oxidant factors in the blood of preeclamptic women. Prenat Diagn. 2009;29:691–696.
[79] Jauniaux E, Watson AL, Hempstock J, Bao YP, Skepper JN, Burton GJ. Onset of maternal arterial blood flow and placental oxidative stress. A possible factor in human early pregnancy failure. Am J Pathol. 2000;157:2111–2122.
[80] Hempstock J, Jauniaux E, Greenwold N, Burton GJ. The contribution of placental oxidative stress to early pregnancy failure. Hum Pathol. 2003;34:1265–1275.
[81] Jauniaux E, Gulbis B, Burton GJ. Physiological implications of the materno-fetal oxygen gradient in human early pregnancy. Reprod Biomed Online. 2003;7:250–253.
[82] Quenby S, Nik H, Innes B, Lash G, Turner M, Drury J, Bulmer J. Uterine natural killer cells and angiogenesis in recurrent reproductive failure. Hum Reprod. 2009;24:45–54.
[83] Burton GJ, Hempstock J, Jauniaux E. Oxygen, early embryonic metabolism and free radical-mediated embryopathies. Reprod Biomed Online. 2003;6:84–96.
[84] Quenby SM, Farquharson RG. Predicting recurring miscarriage: what is important? Obstet Gynecol. 1993;82:132–138.
[85] Rai R, Regan L. Recurrent miscarriage. Lancet. 2006;368:601–611.
[86] Poston L, Raijmakers MT. Trophoblast oxidative stress, antioxidants and pregnancy outcome--a review. Placenta. 2004;25(Suppl A):S72–S78.
[87] Lash GE, Schiessl B, Kirkley M, Innes BA, Cooper A, Searle RF, Robson SC, Bulmer JN. Expression of angiogenic growth factors by uterine natural killer cells during early pregnancy. J Leukoc Biol. 2006;80:572–580.
[88] Hanna J, Goldman-Wohl D, Hamani Y, Avraham I, Greenfield C, Natanson-Yaron S, Prus D, Cohen-Daniel L, Arnon TI, Manaster I. et al. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat Med. 2006;12:1065–1074.
[89] Simsek M, Naziroglu M, Simsek H, Cay M, Aksakal M, Kumru S. Blood plasma levels of lipoperoxides, glutathione peroxidase, beta carotene, vitamin A and E in women with habitual abortion. Cell Biochem Funct. 1998;16:227–231.
[90] Miller H, Wilson R, Jenkins C, MacLean MA, Roberts J, Walker JJ. Glutathione levels and miscarriage. Fertil Steril. 2000;74:1257–1258.
[91] El-Far M, El-Sayed IH, El-Motwally Ael G, Hashem IA, Bakry N. Tumor necrosis factor-alpha and oxidant status are essential participating factors in unexplained recurrent spontaneous abortions. Clin Chem Lab Med. 2007;45:879–883.
[92] Tempfer C, Unfried G, Zeillinger R, Hefler L, Nagele F, Huber JC. Endothelial nitric oxide synthase gene polymorphism in women with idiopathic recurrent miscarriage. Hum Reprod. 2001;16:1644–1647.
[92] Tempfer C, Unfried G, Zeillinger R, Hefler L, Nagele F, Huber JC. Endothelial nitric oxide synthase gene polymorphism in women with idiopathic recurrent miscarriage. Hum Reprod. 2001;16:1644–1647.
[93] Toy H, Camuzcuoglu H, Camuzcuoglu A, Celik H, Aksoy N. Decreased serum prolidase activity and increased oxidative stress in early pregnancy loss. Gynecol Obstet Invest. 2010;69:122–127.
[94] Rumbold A, Duley L, Crowther C, Haslam R. Antioxidants for preventing pre-eclampsia. Cochrane Database Syst Rev. 2005;4:CD004227.
[95] Sata F, Yamada H, Kondo T, Gong Y, Tozaki S, Kobashi G, Kato EH, Fujimoto S, Kishi R. Glutathione S-transferase M1 and T1 polymorphisms and the risk of recurrent pregnancy loss. Mol Hum Reprod. 2003;9:165–169.
[96] Sata F, Yamada H, Yamada A, Kato EH, Kataoka S, Saijo Y, Kondo T, Tamaki J, Minakami H, Kishi R. A polymorphism in the CYP17 gene relates to the risk of recurrent pregnancy loss. Mol Hum Reprod. 2003;9:725–728.
[97] Cotgreave IA. N-acetylcysteine: pharmacological considerations and experimental and clinical applications. Adv Pharmacol. 1997;38:205–227.
[98] Kelly GS. Clinical applications of N-acetylcysteine. Altern Med Rev. 1998;3:114–127.
[98] Kelly GS. Clinical applications of N-acetylcysteine. Altern Med Rev. 1998;3:114–127.
[99] Xu DX, Chen YH, Wang H, Zhao L, Wang JP, Wei W. Effect of N-acetylcysteine on lipopolysaccharide-induced intra-uterine fetal death and intra-uterine growth retardation in mice. Toxicol Sci. 2005;88:525–533.
[100] Amin AF, Shaaban OM, Bediawy MA. N-acetyl cysteine for treatment of recurrent unexplained pregnancy loss. Reprod Biomed Online. 2008;17:722–726
[101] Lappas M, Permezel M, Rice GE. N-Acetyl-cysteine inhibits phospholipid metabolism, proinflammatory cytokine release, protease activity, and nuclear factor-kappaB deoxyribonucleic acid-binding activity in human fetal membranes in vitro. J Clin Endocrinol Metab. 2003;88:1723–1729.
[102] Aluigi MG, De Flora S, D'Agostini F, Albini A, Fassina G. Antiapoptotic and antigenotoxic effects of N-acetylcysteine in human cells of endothelial origin. Anticancer Res. 2000;20:3183–3187.
[103] Kovacic P. Unifying mechanism for addiction and toxicity of abused drugs with application to dopamine and glutamate mediators: electron transfer and reactive oxygen species. Med Hypotheses. 2005;65(1):90-6. doi: 10.1016/j.mehy.2005.01.031. PMID: 15893124.
[104] Hennig B, Hammock BD, Slim R, Toborek M, Saraswathi V, Robertson LW. PCB-induced oxidative stress in endothelial cells: modulation by nutrients. Int J Hyg Environ Health. 2002 Mar;205(1-2):95-102. doi: 10.1078/1438-4639-00134. PMID: 12018021.
[105] Jirsová S, Masata J, Jech L, Zvárová J. Effect of polychlorinated biphenyls (PCBs) and 1,1,1-trichloro-2,2,-bis (4-chlorophenyl)-ethane (DDT) in follicular fluid on the results of in vitro fertilization-embryo transfer (IVF-ET) programs. Fertil Steril. 2010 Apr;93(6):1831-6. doi: 10.1016/j.fertnstert.2008.12.063. Epub 2009 Feb 6. PMID: 19200984.
[106] Samarawickrema N, Pathmeswaran A, Wickremasinghe R, Peiris-John R, Karunaratna M, Buckley N, Dawson A, de Silva J. Fetal effects of environmental exposure of pregnant women to organophosphorus compounds in a rural farming community in Sri Lanka. Clin Toxicol (Phila). 2008 Jul;46(6):489-95. doi: 10.1080/15563650701837030. PMID: 18584359.
[107] Fisher-Wellman K, Bell HK, Bloomer RJ. Oxidative stress and antioxidant defense mechanisms linked to exercise during cardiopulmonary and metabolic disorders. Oxid Med Cell Longev. 2009;2:43–51.
[108] Fisher-Wellman K, Bell HK, Bloomer RJ. Oxidative stress and antioxidant defense mechanisms linked to exercise during cardiopulmonary and metabolic disorders. Oxid Med Cell Longev. 2009;2:43–51.
[109] Fisher-Wellman K, Bloomer RJ. Acute exercise and oxidative stress: a 30 year history. Dyn Med. 2009;8:1
[110] Leelarungrayub D, Saidee K, Pothongsunun P, Pratanaphon S, YanKai A, Bloomer RJ. Six weeks of aerobic dance exercise improves blood oxidative stress status and increases interleukin-2 in previously sedentary women. J Bodyw Mov Ther. 2011;15:355–362.
[111] Fait V, Sela S, Ophir E, Khoury S, Nissimov J, Tkach M, Hirsh Y, Khotaba S, Tarasova L, Oettinger M. Hyperemesis gravidarum is associated with oxidative stress. Am J Perinatol. 2002 Feb;19(2):93-8. doi: 10.1055/s-2002-23554. PMID: 11938483.
[112] Holthe MR, Staff AC, Berge LN, Lyberg T. Leukocyte adhesion molecules and reactive oxygen species in preeclampsia. Obstet Gynecol. 2004 May;103(5 Pt 1):913-22. doi: 10.1097/01.AOG.0000124806.39111.ba. PMID: 15121565.
[113] Diamant S, Kissilevitz R, Diamant Y. Lipid peroxidation system in human placental tissue: general properties and the influence of gestational age. Biol Reprod. 1980 Nov;23(4):776-81. doi: 10.1095/biolreprod23.4.776. PMID: 7448277.
[114] Little RE, Gladen BC. Levels of lipid peroxides in uncomplicated pregnancy: a review of the literature. Reprod Toxicol. 1999 Sep-Oct;13(5):347-52. doi: 10.1016/s0890-6238(99)00033-7. PMID: 10560582.
[115] Myatt L, Cui X. Oxidative stress in the placenta. Histochem Cell Biol. 2004 Oct;122(4):369-82. doi: 10.1007/s00418-004-0677-x. Epub 2004 Jul 10. PMID: 15248072.
[116] Wang YP, Walsh SW, Guo JD, Zhang JY. Maternal levels of prostacyclin, thromboxane, vitamin E, and lipid peroxides throughout normal pregnancy. Am J Obstet Gynecol. 1991 Dec;165(6 Pt 1):1690-4. doi: 10.1016/0002-9378(91)90016-k. PMID: 1750461.
[117] Gupta S, Aziz N, Sekhon L, Agarwal R, Mansour G, Li J, Agarwal A. Lipid peroxidation and antioxidant status in preeclampsia: a systematic review. Obstet Gynecol Surv. 2009;64:750–759.
[118] Reslan OM, Khalil RA. Molecular and vascular targets in the pathogenesis and management of the hypertension associated with preeclampsia. Cardiovasc Hematol Agents Med Chem. 2010;8:204–226.
[119] Roberts JM, Pearson GD, Cutler JA, Lindheimer MD. Summary of the NHLBI Working Group on Research on Hypertension During Pregnancy. Hypertens Pregnancy. 2003;22:109–127.
[120] National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000;183:S1–S22.
[121] American College of Obstetricians and Gynecologists. Practice bulletin No.33: Diagnosis and management of preeclampsia and eclampsia. Obstet Gynecol. 2002;99:159–167.
[122] Myatt L, Cui X. Oxidative stress in the placenta. Histochem Cell Biol. 2004;122:369–382.
[123] Khalil RA, Granger JP. Vascular mechanisms of increased arterial pressure in preeclampsia: lessons from animal models. Am J Physiol Regul Integr Comp Physiol. 2002;283:R29–R45.
[124] Burton GJ, Jauniaux E. Placental oxidative stress: from miscarriage to preeclampsia. J Soc Gynecol Investig. 2004;11:342–352.
[125] Redman CW, Sargent IL. Placental stress and pre-eclampsia: a revised view. Placenta. 2009;30(Suppl A):S38–S42.
[126] Walsh SW. Eicosanoids in preeclampsia. Prostaglandins Leukot Essent Fatty Acids. 2004;70:223–232.
[127]Klemmensen A, Tabor A, Osterdal ML, Knudsen VK, Halldorsson TI, Mikkelsen TB, Olsen SF. Intake of vitamin C and E in pregnancy and risk of pre-eclampsia: prospective study among 57 346 women. BJOG. 2009;116:964–974.
[128] Bodnar LM, Tang G, Ness RB, Harger G, Roberts JM. Periconceptional multivitamin use reduces the risk of preeclampsia. Am J Epidemiol. 2006;164:470–477.
[129] Catov JM, Nohr EA, Bodnar LM, Knudson VK, Olsen SF, Olsen J. Association of periconceptional multivitamin use with reduced risk of preeclampsia among normal-weight women in the Danish National Birth Cohort. Am J Epidemiol. 2009;169:1304–1311.
[130] Bloomer RJ, Fisher-Wellman KH. Systemic oxidative stress is increased to a greater degree in young, obese women following consumption of a high fat meal. Oxid Med Cell Longev. 2009 Jan-Mar;2(1):19-25. doi: 10.4161/oxim.2.1.7860. PMID: 20046641; PMCID: PMC2763227.
[131] Mistry HD, Williams PJ. The importance of antioxidant micronutrients in pregnancy. Oxid Med Cell Longev. 2011;2011:841749. doi: 10.1155/2011/841749. Epub 2011 Sep 13. PMID: 21918714; PMCID: PMC3171895.
[132] Robker RL, Akison LK, Bennett BD, Thrupp PN, Chura LR, Russell DL, Lane M, Norman RJ. Obese women exhibit differences in ovarian metabolites, hormones, and gene expression compared with moderate-weight women. J Clin Endocrinol Metab. 2009 May;94(5):1533-40. doi: 10.1210/jc.2008-2648. Epub 2009 Feb 17. PMID: 19223519.
[133] Higashi Y, Sasaki S, Nakagawa K, Kimura M, Noma K, Hara K, Matsuura H, Goto C, Oshima T, Chayama K. et al. Low body mass index is a risk factor for impaired endothelium-dependent vasodilation in humans: role of nitric oxide and oxidative stress. J Am Coll Cardiol. 2003;42:256–263.
[134] Khan RN, Matharoo-Ball B, Shaw RW. Antioxidant enzyme expression, lipid peroxidation, and protein oxidation in human myometrium with parturition. Reprod Sci. 2010;17:78–84.
[135] Mustafa MD, Pathak R, Ahmed T, Ahmed RS, Tripathi AK, Guleria K, Banerjee BD. Association of glutathione S-transferase M1 and T1 gene polymorphisms and oxidative stress markers in preterm labor. Clin Biochem. 2010;43:1124–1128.
[136] Pathak R, Suke SG, Ahmed T, Ahmed RS, Tripathi AK, Guleria K, Sharma CS, Makhijani SD, Banerjee BD. Organochlorine pesticide residue levels and oxidative stress in preterm delivery cases. Hum Exp Toxicol. 2010;29:351–358.
[137] Hong YC, Lee KH, Yi CH, Ha EH, Christiani DC. Genetic susceptibility of term pregnant women to oxidative damage. Toxicol Lett. 2002;129:255–262.
[138] Frosali S, Di Simplicio P, Perrone S, Di Giuseppe D, Longini M, Tanganelli D, Buonocore G. Glutathione recycling and antioxidant enzyme activities in erythrocytes of term and preterm newborns at birth. Biol Neonate. 2004;85:188–194.
[139] Mistry HD, Williams PJ. The importance of antioxidant micronutrients in pregnancy. Oxid Med Cell Longev. 2011;2011:841749. doi: 10.1155/2011/841749. Epub 2011 Sep 13. PMID: 21918714; PMCID: PMC3171895. https://pubmed.ncbi.nlm.nih.gov/21918714/#&gid=article-figures&pid=figure-1-uid-0. Accessed 22Jul23
[140] Boutros T, Chevet E, Metrakos P. Mitogen-activated protein (MAP) kinase/MAP kinase phosphatase regulation: roles in cell growth, death, and cancer. Pharmacol Rev. 2008;60:261–310.
[141] Miller H, Wilson R, Jenkins C, MacLean MA, Roberts J, Walker JJ. Glutathione levels and miscarriage. Fertil Steril. 2000;74:1257–1258.
[142] Burton GJ, Jauniaux E. Placental oxidative stress: from miscarriage to preeclampsia. J Soc Gynecol Investig. 2004;11:342–352.
[143] Redman CW, Sargent IL. Placental stress and pre-eclampsia: a revised view. Placenta. 2009;30(Suppl A):S38–S42.
[144] Burton GJ, Yung HW, Cindrova-Davies T, Charnock-Jones DS. Placental endoplasmic reticulum stress and oxidative stress in the pathophysiology of unexplained intrauterine growth restriction and early onset preeclampsia. Placenta. 2009;30(Suppl A):S43–S48.
[145] Kharfi A, Giguere Y, De Grandpre P, Moutquin JM, Forest JC. Human chorionic gonadotropin (hCG) may be a marker of systemic oxidative stress in normotensive and preeclamptic term pregnancies. Clin Biochem. 2005;38:717–721.
[146] Roberts JM, Cooper DW. Pathogenesis and genetics of pre-eclampsia. Lancet. 2001;357:53–56.
[147] Granger JP, Alexander BT, Llinas MT, Bennett WA, Khalil RA. Pathophysiology of preeclampsia: linking placental ischemia/hypoxia with microvascular dysfunction. Microcirculation. 2002;9:147–160.
[148] Hung TH, Skepper JN, Burton GJ. In vitro ischemia-reperfusion injury in term human placenta as a model for oxidative stress in pathological pregnancies. Am J Pathol. 2001;159:1031–1043.
[[149] Irani K. Oxidant signaling in vascular cell growth, death, and survival: a review of the roles of reactive oxygen species in smooth muscle and endothelial cell mitogenic and apoptotic signaling. Circ Res. 2000;87:179–183.
[150] Aris A, Benali S, Ouellet A, Moutquin JM, Leblanc S. Potential biomarkers of preeclampsia: inverse correlation between hydrogen peroxide and nitric oxide early in maternal circulation and at term in placenta of women with preeclampsia. Placenta. 2009;30:342–347.
[151] Sharma JB, Sharma A, Bahadur A, Vimala N, Satyam A, Mittal S. Oxidative stress markers and antioxidant levels in normal pregnancy and pre-eclampsia. Int J Gynaecol Obstet. 2006;94:23–27.
[152] Haque SK, Siddiqui MU, Islam N, Moin S. Erythrocyte markers of oxidative stress in higher age-group preeclamptic and normal pregnant mothers. Hypertens Pregnancy. 2010;29:69–81.
[153] Padmini E, Lavanya S, Uthra V. Preeclamptic placental stress and over expression of mitochondrial HSP70. Clin Chem Lab Med. 2009;47:1073–1080.
[154] Tsukimori K, Komatsu H, Fukushima K, Kaku T, Nakano H, Wake N. Inhibition of nitric oxide synthetase at mid-gestation in rats is associated with increases in arterial pressure, serum tumor necrosis factor-alpha, and placental apoptosis. Am J Hypertens. 2008;21:477–481.
[155] Bhatnagar S, Bhattacharjee J, Vaid M, Madan T, Trivedi SS, Sarma PU. Inducible nitric oxide synthase (iNOS) gene polymorphism in pre-eclampsia: a pilot study in North India. Aust N Z J Obstet Gynaecol. 2007;47:477–482.
[156] Vural P. Nitric oxide/endothelin-1 in preeclampsia. Clin Chim Acta. 2002;317:65–70.
[157] Cindrova-Davies T. Gabor Than Award Lecture 2008: pre-eclampsia - from placental oxidative stress to maternal endothelial dysfunction. Placenta. 2009;30(Suppl A):S55–S65.
[158] Levine RJ, Maynard SE, Qian C, Lim KH, England LJ, Yu KF, Schisterman EF, Thadhani R, Sachs BP, Epstein FH. et al. Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med. 2004;350:672–683.
[159] Ahmed A, Rahman M, Zhang X, Acevedo CH, Nijjar S, Rushton I, Bussolati B, St John J. Induction of placental heme oxygenase-1 is protective against TNFalpha-induced cytotoxicity and promotes vessel relaxation. Mol Med. 2000;6:391–409.
[160] De Marco CS, Caniggia I. Mechanisms of oxygen sensing in human trophoblast cells. Placenta. 2002;23:S58–S68.
[161] Li H, Gu B, Zhang Y, Lewis DF, Wang Y. Hypoxia-induced increase in soluble Flt-1 production correlates with enhanced oxidative stress in trophoblast cells from the human placenta. Placenta. 2005;26:210–217.
[162] Farina A, Sekizawa A, De Sanctis P, Purwosunu Y, Okai T, Cha DH, Kang JH, Vicenzi C, Tempesta A, Wibowo N. et al. Gene expression in chorionic villous samples at 11 weeks' gestation from women destined to develop preeclampsia. Prenat Diagn. 2008;28:956–961.
[163] Griendling KK, Sorescu D, Ushio-Fukai M. NAD(P)H oxidase: role in cardiovascular biology and disease. Circ Res. 2000;86:494–501.
[164] Wallekut G, Homuth V, Fischer T, Linschau C, Horstkamp B, Jüpner A, VBaur E, Nissen E, Vetter K, Neichel D. et al. Patients with preeclampsia develop agonistic autoantibodies against the angiotensin AT1 receptor. J Clin Invest. 1999;103:945–952.
[165] Dechend R, Viedt C, Muller DN, Ugele B, Brandes RP, Wallukat G, Park JK, Janke J, Barta P, Theuer J. et al. AT1 receptor agonistic antibodies from preeclamptic patients stimulate NADPH oxidase. Circulation. 2003;107:1632–1639.
[166] Raijmakers MT, Peters WH, Steegers EA, Poston L. NAD(P)H oxidase associated superoxide production in human placenta from normotensive and pre-eclamptic women. Placenta. 2004;25(Suppl A):S85–S89.
[167] Griendling KK, Sorescu D, Lassegue B, Ushio-Fukai M. Modulation of protein kinase activity and gene expression by reactive oxygen species and their role in vascular physiology and pathophysiology. Arterioscler Thromb Vasc Biol. 2000;20:2175–2183.
[168] Costa LG, Vitalone A, Cole TB, Furlong CE. Modulation of paraoxonase (PON1) activity. Biochem Pharmacol. 2005;69:541–550.
[169] Baker AM, Klein RL, Haeri S, Moss KL, Boggess KA. Association of Midgestational Paraoxonase 1 activity with pregnancies complicated by preeclampsia. Am J Perinatol. 2010;27:205–210.
[170] Kumru S, Aydin S, Gursu MF, Ozcan Z. Changes of serum paraoxonase (an HDL-cholesterol-associated lipophilic antioxidant) and arylesterase activities in severe preeclamptic women. Eur J Obstet Gynecol Reprod Biol. 2004;114:177–181.
[171] Uzun H, Benian A, Madazli R, Topcuoglu MA, Aydin S, Albayrak M. Circulating oxidized low-density lipoprotein and paraoxonase activity in preeclampsia. Gynecol Obstet Invest. 2005;60:195–200.
[172] Biri A, Bozkurt N, Turp A, Kavutcu M, Himmetoglu O, Durak I. Role of oxidative stress in intrauterine growth restriction. Gynecol Obstet Invest. 2007;64:187–192.
[173] Karowicz-Bilinska A. Lipid peroxides concentration in women with intrauterine growth restriction. Ginekol Pol. 2004;75:6–9.
[174] Potdar N, Singh R, Mistry V, Evans MD, Farmer PB, Konje JC, Cooke MS. First-trimester increase in oxidative stress and risk of small-for-gestational-age fetus. BJOG. 2009;116:637–642.
[175] Levy R, Smith SD, Chandler K, Sadovsky Y, Nelson DM. Apoptosis in human cultured trophoblasts is enhanced by hypoxia and diminished by epidermal growth factor. Am J Physiol Cell Physiol. 2000;278:C982–C988.
[176] Levy R, Smith SD, Yusuf K, Huettner PC, Kraus FT, Sadovsky Y, Nelson DM. Trophoblast apoptosis from pregnancies complicated by fetal growth restriction is associated with enhanced p53 expression. Am J Obstet Gynecol. 2002;186:1056–1061.
[177] Heazell AE, Lacey HA, Jones CJ, Huppertz B, Baker PN, Crocker IP. Effects of oxygen on cell turnover and expression of regulators of apoptosis in human placental trophoblast. Placenta. 2008;29:175–186.
[178] Hung TH, Burton GJ. Hypoxia and reoxygenation: a possible mechanism for placental oxidative stress in preeclampsia. Taiwan J Obstet Gynecol. 2006;45:189–200.
[179] Yung HW, Calabrese S, Hynx D, Hemmings BA, Cetin I, Charnock-Jones DS, Burton GJ. Evidence of placental translation inhibition and endoplasmic reticulum stress in the etiology of human intrauterine growth restriction. Am J Pathol. 2008;173:451–462.
[180] Hafner E, Metzenbauer M, Hofinger D, Munkel M, Gassner R, Schuchter K, Dillinger-Paller B, Philipp K. Placental growth from the first to the second trimester of pregnancy in SGA-foetuses and pre-eclamptic pregnancies compared to normal foetuses. Placenta. 2003;24:336–342.
[181] Pezaro S, Pearce G, and Reinhold E. Hypermobile Ehlers-Danlos Syndrome during pregnancy, birth and beyond. British Journal of Midwifery. 2018; 26(4): 217-223.
[182] Salmon AB, Richardson A, Pérez VI. Update on the oxidative stress theory of aging: does oxidative stress play a role in aging or healthy aging? Free Radic Biol Med. 2010 Mar 1;48(5):642-55. doi: 10.1016/j.freeradbiomed.2009.12.015. Epub 2009 Dec 28. PMID: 20036736; PMCID: PMC2819595.
[183] Crist BL, Alekel DL, Ritland LM, Hanson LN, Genschel U, Reddy MB. Association of oxidative stress, iron, and centralized fat mass in healthy postmenopausal women. J Womens Health (Larchmt). 2009 Jun;18(6):795-801. doi: 10.1089/jwh.2008.0988. PMID: 19456248; PMCID: PMC2835408.
[184] Tropea A, Miceli F, Minici F, Tiberi F, Orlando M, Gangale MF, Romani F, Catino S, Mancuso S, Navarra P, Lanzone A, Apa R. Regulation of vascular endothelial growth factor synthesis and release by human luteal cells in vitro. J Clin Endocrinol Metab. 2006 Jun;91(6):2303-9. doi: 10.1210/jc.2005-2457. Epub 2006 Apr 4. PMID: 16595603.
[185] Chandankhede M, Gupta M, Pakhmode S. Assessment of Psychological Status and Oxidative Stress in Postmenopausal Women: A Cross-Sectional Study. J Menopausal Med. 2021 Dec;27(3):155-161. doi: 10.6118/jmm.20035. PMID: 34989189; PMCID: PMC8738848.
[186] Broekmans FJ, Soules MR, Fauser BC. Ovarian aging: mechanisms and clinical consequences. Endocr Rev. 2009;30:465–493.
[187] Bromberger JT, Kravitz HM, Chang Y, Randolph JF, Jr, Avis NE, Gold EB, et al. Does risk for anxiety increase during the menopausal transition? Study of women's health across the nation. Menopause. 2013;20:488–495.
[188] Enns DL, Tiidus PM. The influence of estrogen on skeletal muscle: sex matters. Sports Med. 2010;40:41–58.
[189] Kumar S, Lata K, Mukhopadhyay S, Mukherjee TK. Role of estrogen receptors in pro-oxidative and anti-oxidative actions of estrogens: a perspective. Biochim Biophys Acta. 2010;1800:1127–1135.
[190] Doshi SB, Agarwal A. The role of oxidative stress in menopause. J Midlife Health. 2013;4:140–146.
[191] Malacara JM. The psychosocial factors in menopause. Rev Endocrinol Nutr. 2006;14:137–140.
[192] Liu ZM, Ho SC, Xie YJ, Chen YJ, Chen YM, Chen B, et al. Associations between dietary patterns and psychological factors: a cross-sectional study among Chinese postmenopausal women. Menopause. 2016;23:1294–1302.
[193] Abshirini M, Siassi F, Koohdani F, Qorbani M, Khosravi S, Hedayati M, et al. Dietary total antioxidant capacity is inversely related to menopausal symptoms: a cross-sectional study among Iranian postmenopausal women. Nutrition. 2018;55-56:161–167.
[194] Landucci, E., Laurino, A., Cinci, L., Gencarelli, M., & Raimondi, L. (2019). Thyroid Hormone, Thyroid Hormone Metabolites and Mast Cells: A Less Explored Issue. Frontiers in cellular neuroscience, 13, 79. doi:10.3389/fncel.2019.00079.
Testimonials

Working with this life coach has been a game-changer for me. Their guidance helped me gain clarity on my goals, overcome self-doubt, and take decisive action in my career and personal life. I now feel more confident, focused, and motivated to achieve my dreams. I highly recommend this coaching service to anyone looking for real, lasting transformation.
John D


I never realized how much a life coach could help until I started working with this team. The support and actionable advice I’ve received have transformed the way I approach challenges and set goals. I’m more confident in my decisions and have seen incredible growth in both my career and personal life. Truly an investment in myself.
Sarah M

Shop: 25 Highspire Rd, Richboro PA 18954
Call (445) 455-4600
Email: [email protected]
Site: https//:soimt.org
